
An ideal gas is made to undergo the
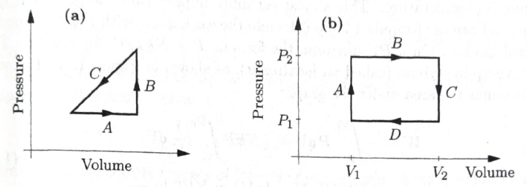
Figure 1.10. PV diagrams for Problem 1.33 and 1.34.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
An Introduction to Thermal Physics
Additional Science Textbook Solutions
Conceptual Integrated Science
College Physics: A Strategic Approach (4th Edition)
Essential University Physics: Volume 2 (3rd Edition)
Physics for Scientists and Engineers with Modern Physics
Essential University Physics: Volume 1 (3rd Edition)
College Physics (10th Edition)
- Consider the thermodynamic process, A->B->C->A shown above. The heat absorbed during A->B is 591J. If the change in internal energy during B->C is 4146J, What is the change in internal energy in SI units during C->A? Express only the number of your answer with 4 significant figures.arrow_forwardThe figure (Figure 1)shows the cycle for a heat engine that uses a gas having γ=1.25. The initial temperature is T1=300K, and this engine operates at 30 cycles per second. What is the power output of the engine? Pout = 1.6 kW What is the engine's thermal efficiency?arrow_forwardA container is filled with an ideal diatomic gas to a pressure and volume of P₁ and V₁, respectively. The gas is then warmed in a two-step process that increases the pressure by a factor of four and the volume by a factor of five. Determine the amount of energy transferred to the gas by heat if the first step is carried out at constant volume and the second step at constant pressure. (Use any variable or symbol stated above as necessary.) Q =arrow_forward
- Converting sunlight to electricity with solar cells has an efficiency of 15%. It's possible to achieve a higher efficiency (though currently at higher cost) by using concentrated sunlight as the hot reservoir of a heat engine. Each dish in (Figure 1) concentrates sunlight on one side of a heat engine, producing a hot-reservoir temperature of 560 ∘C. The cold reservoir, ambient air, is approximately 30 ∘C. The actual working efficiency of this device is 30%. What is the theoretical maximum efficiency?arrow_forwardA quantity of a monatomic ideal gas undergoes a process in which both its pressure and volume are increased by a factor of n = 9 as shown in the figure below. What is the energy absorbed by heat into the gas during this process? Hint: The internal energy of a monatomic ideal gas at pressure P and occupying volume V is given by U = 3PV. 2 (Give your answer as a multiple of Povo.) x Your response differs significantly from the correct answer. Rework your solution from the beginning and check each step carefully. x PoVo P nPo 8 Po Vo nVo Varrow_forwardThe temperature at state A is 20°C, that is 293 K. What is the heat (Q) for process A to D, in MJ (MegaJoules)? (Hint: What is the change in thermal energy and work done by the gas for this process?) Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. P (atm) A. 4 2 V (m) 2 3 4 5arrow_forward
- A gas expands from I to F in the figure below. The energy added to the gas by heat is 276 J when the gas goes from I to F along the diagonal path. A pressure-volume graph consists of points and line segments plotted on a coordinate plane, where the horizontal axis is V (liters)and the vertical axis is P (atm). Three points are plotted: point I at (2, 4) point A at (4, 4) point F at (4, 1) Line segments connect the three points to form a triangle. Arrows along the line segments point from I to A, from A to F, and from I to F. (a) What is the change in internal energy of the gas? J (b) How much energy must be added to the gas by heat along the indirect path IAF?arrow_forwardPart A: If you supply 2950 J of heat to 6.00 moles of an ideal diatomic gas initially at 23.0 ∘C in a perfectly rigid container, what will be the final temperature of the gas? Express in degrees Celsius. Part B: Suppose the gas in the container were an ideal monatomic gas instead. How much heat would you need to add to produce the same temperature change? Part C: Which pV diagram expresses these processes?arrow_forwardCan you help 2a and subpart 2b 1. A certain power plant produces useful work (i.e. electricity) at a rate of 900 MW (remember, that's 900 x 106 joules/sec). How many joules of electricity does this power plant produce in 24 hours? 2a. To generate this amount of electricity, the power plant uses 2.22 x 1014 J of heat drawn from burning coal. What is the efficiency of this power plant? 2b. How much waste heat, Qc, does this power plant eject into its cold reservoir in 24 hoursarrow_forward
- Please describe to me the laws of thermodynamics. There are a certain number of them and therefore you must have all of them. Each should be labeled with it's number. For instance "The First Law of thermodynamics says that..."arrow_forwardThe temperature at state A is 20°C, that is 293 K. What is the heat (Q) for process D to B, in MJ (MegaJoules)? (Hint: What is the change in thermal energy and work done by the gas for this process?) Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. p (atm) 4 3 2 -> +V (m) 5arrow_forwardThe figure (Figure 1)shows the cycle for a heat engine that uses a gas having y = 1.25. The initial temperature is T1= 300 K, and this engine operates at 16 cycles per second.arrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





