
Concept explainers
Draw the structure consistent with each description.
a.
b.
c.
(a)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The prefix
Answer to Problem 16.10P
The structure corresponding to
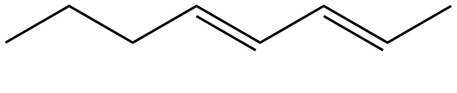
Explanation of Solution
The given name is
Thus, the correct structure of
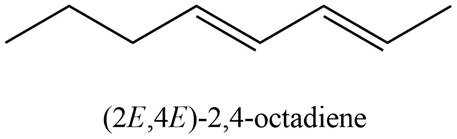
Figure 1
The structure corresponding to
(b)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The prefix
Answer to Problem 16.10P
The structure corresponding to
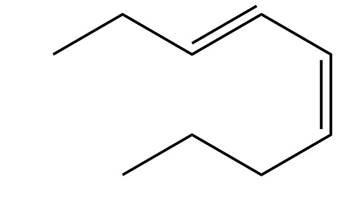
Explanation of Solution
The given name is
Thus, the correct structure of
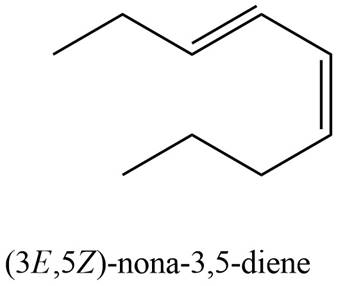
Figure 2
The structure corresponding to
(c)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The prefix
Answer to Problem 16.10P
The structures corresponding to
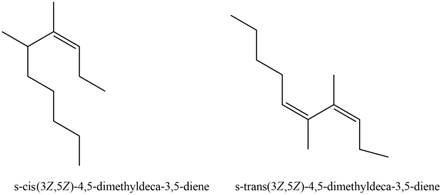
Explanation of Solution
The given name is
Thus, the correct structure to
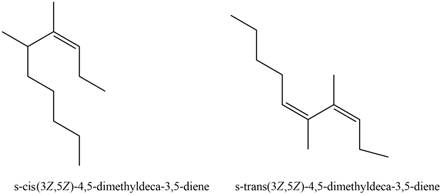
Figure 3
The structure corresponding to
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry
- A B C D E 1. NaBH4 2. H3O+ 1. CH3MgBr 2. H3O+ 1. (CH3)2CuLi 2. H3O+ 1. CH3Li 2. H3O+ 1. (CH3CH2)2CuLi 2. H3O+arrow_forwardDraw the structure consistent with each description. a.(2E,4E)-octa-2,4-diene in the s-trans conformation b.(3E,5Z)-nona-3,5-diene in the s-cis conformation c.(3Z,5Z)-4,5-dimethyldeca-3,5-diene. Draw both the s-cis and s-trans conformations.arrow_forwardcyclopentene + NBS/ CCI4/ light --> O (1R, 2R)-dibromocyclopentene + enantiomer O 3-bromocyclopentene O 1,2-dibromocyclopentene O 1-bromo-2-methylcyclopentanearrow_forward
- Order the following in increasing priority A. -C, -CH, -OL B. -CH3, -CH2OH, -CH2CH3 C. -C≡CH, -CH゠CH2, -CH゠Oarrow_forward5. Chairs and E2 b. a. Draw the line-bond structure of (1R, 2S,3S)-1-ethyl-2-iodo-3-isopropylcyclohexane. Draw both chair flips of (1R, 2S,3S)-1-ethyl-2-iodo-3-isopropylcyclohexane. Showing all calculations, determine which is the more stable conformation. C. Which chair flip is able to undergo E2 elimination? Justify your answer. d. Show the product of such an E2 elimination using NaOEt as base.arrow_forward3) Draw the ECSF structures of (CH3)3CO (tert-butoxide ion) and CH30º (methoxide ion). Why is (tert-butoxide a PN but methoxide a GN even though both have a full O? (As it turns out, both are SBs) Redraw (CH3)3coe (ECSF) Why PN? Redraw CH30S (ECSF) Why GN?arrow_forward
- Revision-Chapter 10 – Reactions in aldehyde and ketones Complete the following reaction equation. a) CH=C 1) 0, H3C-HC CH, CH3 CH3 2) Zn/ H,0 b) PCC CH;Cl2 c) CI -CH3 AICI CH3 R CH2. d) H,0* KMno, H3C H3C *CH2arrow_forwardSet C. Some Relatively Easy Molecules (1) SF;CI (2) Chlorobenzene (3) 1,2-Dichlorobenzene (4) 1,3-Dichlorobenzene (5) 1,4-Dichlorobenzene (6) 1,3,5-Trichlorobenzene (7) 1,2,3-Trichlorobenzene (8) 1,2,4-Trichlorobenzene (9) trans-(CrCl,(H,O).J* (ignore H atoms) (10) (11) OPCI, (12) trans-Pt(NH,),Cl; (ignore H atoms) (13) cis-Pt(NH,),Cl, (ignore H atoms) (14) BICIF (15) What is the point group for each of the following substituted cyclo- butanes? Assume that C,H, itself has D symmetry and that replacing an H by X or Y changes no other structure parameters. (a) (d)arrow_forward4. Ring Inversion of Substituted Cyclohexane 4.1 Two Chair Conformations of Substituted Cyclohexane Rings boat conformation is 30 hair conformation. Thus, the equilibrium favors the chair conformations. _kJ/mol higher ● ● When Ring Flipping Occurs If a substituent is initially in the axial position, after ring flipping it will be in the position (and vice-versa). equatorial 4 5 5 Connectivity does NOT change. Substituents stay on the same side of the ring that they started on! The cis-trans relationships do NOT change (cis groups stay cis and trans groups stay trans.) H3C Pown 6 3 3 6 CH3 1 CH3 2 1 2 eq I I CH3 H 180N -I 6 in energy than the down ax CH₂ On up on C 1arrow_forward
- O O O H CH3 OH H H H H OH H H H 安安安安 H OH H HO H CH(CH3)2 CH(CH3)2 CH(CH3)2 CH(CH3)2 A C Which C2-C3 Newman projection represents (S)-4-methylpentan-2-ol? m A None of these OH .... (S)-4-methylpentan-2-ol B C Darrow_forward6) Which staggered Newman Projection(s), looking down the C2 - C3 bond (C2 in front and C3 in back), illustrates the following boxed compound? OH CH3 H3C H CH3 .H CH=CH2 но. H3C. он но ČH=CH2 H. H3C H. ČH3 ČH=CH2 II III CH3 CH3 CH3 H. CH3 но H. OH ČH=CH2 ČH=CH2 IV Varrow_forward2. Convert the line structure of 2,3-dimethylpentane into a Newman projection as viewed along the C2-C3 bond (C2 in front, C3 in back). Draw the most stable conformation. Two templates are given with one hydrogen atom drawn in place. Note that only one template leads to the correct answer, therefore use only one of the given templates. t Н. H.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
