
(a)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
(1) The six-membered aromatic ring is named as benzene.
(2) If a
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 1, the benzene ring contains chloro and the ethyl group. The numbering starts from the carbon atom containing chloro group since it comes first in the alphabetical series and the numbering is done according to lowest locant rule.
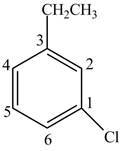
Figure 1
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(b)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 2, the benzene ring contains two ethyl groups. So, numbering can be done from any carbon containing ethyl group using lowest locant rule.
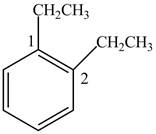
Figure 2
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(c)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 3, the benzene ring contains nitro and the vinyl group. The numbering starts from the carbon atom containing nitrro group since it comes first in the alphabetical series and the numbering is done according to lowest locant rule.
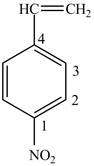
Figure 3
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(d)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 4, the benzene ring contains two chloro groups and one functional group, hydroxyl group. The name of the benzene ring will become phenyl. The numbering starts from the carbon atom containing hydroxyl group since it is a functional group and then the substituents are numbered using lowest locant rule.
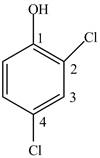
Figure 4
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(e)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 5, the benzene ring contains chloro, bromo, fluoro, iodo and nitro groups. The numbering is done using the lowest locant rule.
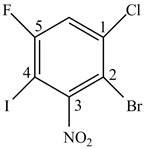
Figure 5
The rule of naming an aromatic compound according to IUPAC is
The IUPAC name of the aromatic compound is
(f)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
(6) If benzene ring is a substitutent, it is named as phenyl.
In Figure 6, the benzene rings are subtituents on the methyl group.
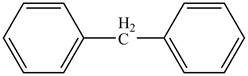
Figure 6
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry
- Are the following halogenated or non- halogented?(a) Cyclohexyl hydrogen sulfate(b) p-Toluenesulfonic acidarrow_forward(a) Draw the structures of the following compounds :(i) 4-Chloropentan-2-one (ii) p-Nitropropiophenone(b) Give tests to distinguish between the following pairs of compounds :(i) Ethanal and Propanal (ii) Phenol and Benzoic acid(iii) Benzaldehyde and Acetophenonearrow_forwardWhat will be the color of the flame and the amount of soot if the following are ignited:(a) Hexane (b)Heptane(c) Cyclohexane (d) Cyclohexene (e) Benzene (f) Toluenearrow_forward
- Propose structures for molecules that fit the following descriptions:(a) An aldehyde with the formula C5H10O(b) An ester with the formula C6H12O2(c) A compound with the formula C3H7NOS that is both anamide and a thiolarrow_forward(a) Give chemical tests to distinguish between the following :(i) Benzoic acid and ethyl benzoate (ii) Benzaldehyde and acetophenonearrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward
- (c) Arrange the following compounds in order of increasing acidity, and explain the reasons for your choice of order: phenol, cyclohexanol, 2-fluorocyclohexanol, 2-fluorophenol.arrow_forward(i) State reagents G and J. (ii) Draw the structural formula for compounds D, E and H.arrow_forward(a) Draw the structure of the following :(i) p-Methylbenzaldehyde (ii) 4-Methylpent-3-en-2-one(b) Give chemical tests to distinguish between the following pairs of compounds :(i) Benzoic acid and Ethyl benzoate, (ii) Benzaldehyde and Acetophenone.(iii) Phenol and Benzoic acid.arrow_forward
- Explain the following statements. You must use chemical equations to justify your explanation. (ii) (I) Phenol is more acidic than cyclohexanol.arrow_forward(a) Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Give two reasons.(b) How will you bring about the following converstions?(i) Propanone to propane (ii) Benzoyl chloride to benzaldehyde(iii) Ethanal to but-2-enalarrow_forwardGive reasons: (i) Bond length of C = O in carboxylic acids is slightly larger than C = O bond length in carbonyl compounds. (ii) There are two –NH2 groups in semicarbazide. However, only one –NH2 group is involved in the formation of semicarbazones. (iii) Benzoic acid is less soluble in water than acetic acid. (iv) Formic acid is a stronger acid than acetic acid.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





