
Concept explainers
(a)
Interpretation:
The IUPAC name for the ester formed when ethanoic acid and propyl alcohol react has to be assigned.
Concept Introduction:
Esters are prepared by condensation of
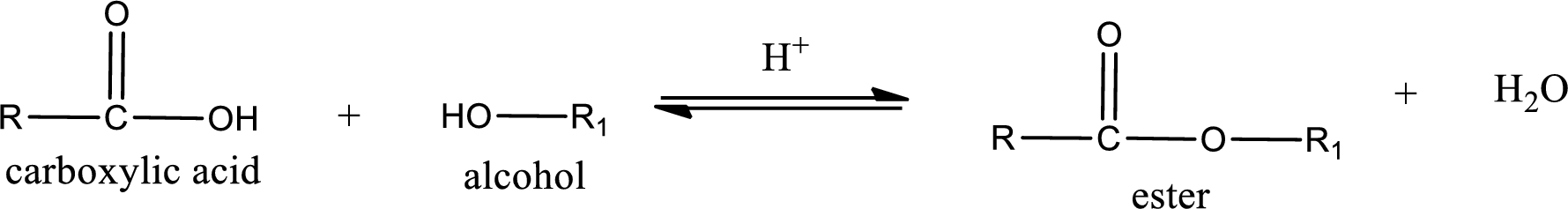
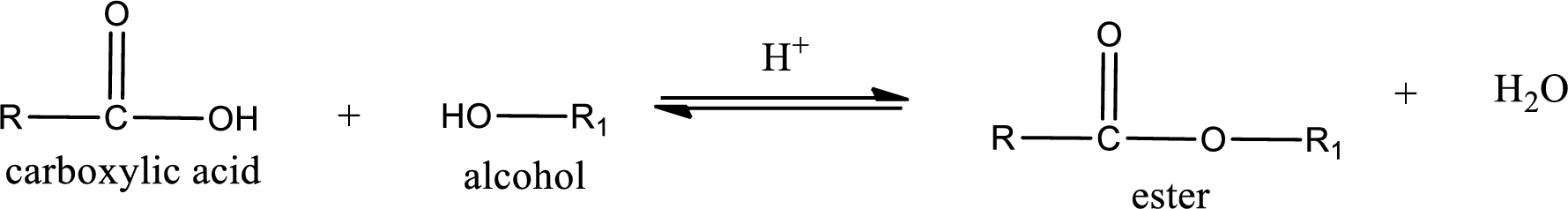
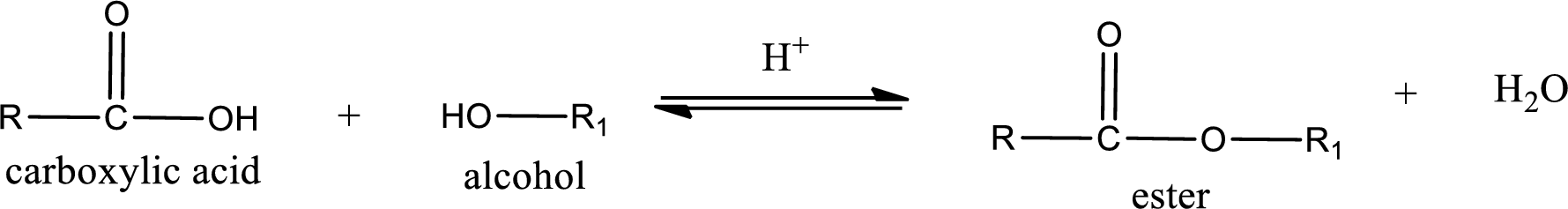
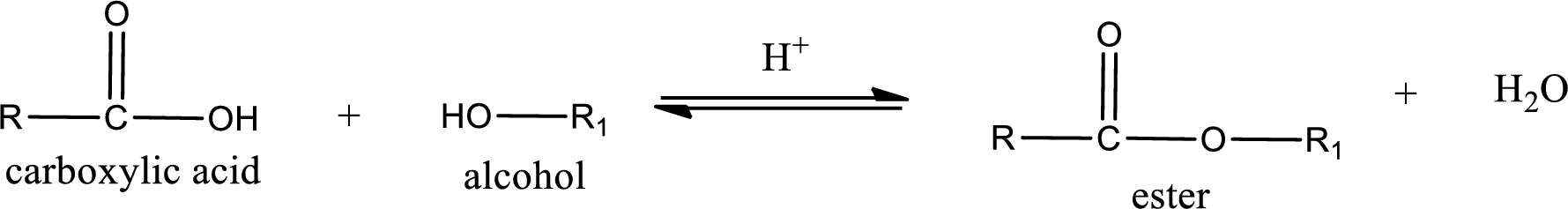
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
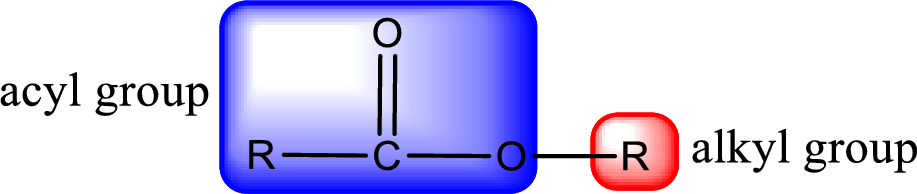
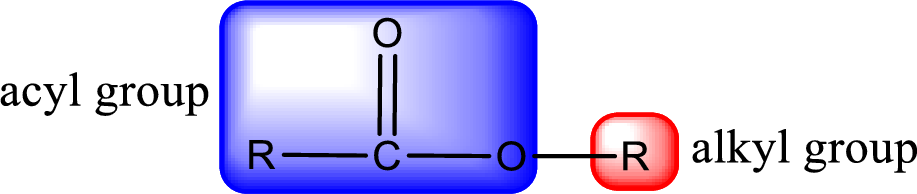
For naming an ester, it can be structurally viewed in a way that contains an acyl group and an alkyl group.
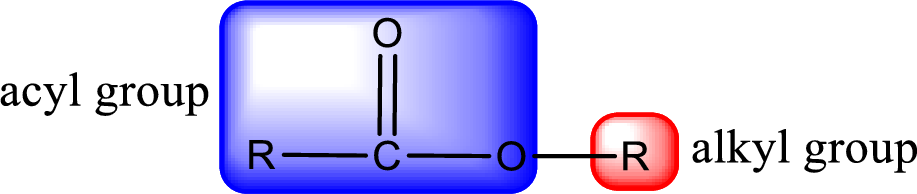
Rules to obtain IUPAC name and common name for an ester:
- Alkyl part appears first in the IUPAC name and it is followed by the acyl part of ester as a separate word.
- Name of the alkyl part in the ester is just a name of R group. It can be alkyl, cycloalkyl, or aryl group.
- Acyl part present in the ester is named by considering the acid name and replacing the suffix “-ic acid” with “-ate”.
- To obtain the common name the alkyl part name is the same while the acyl part name is derived from the common name of the acid by replacing the suffix “-ic acid” with “-ate”.
(a)
Answer to Problem 16.98EP
IUPAC name of the ester formed is propyl ethanoate.
Explanation of Solution
Given carboxylic acid and alcohol structure is,

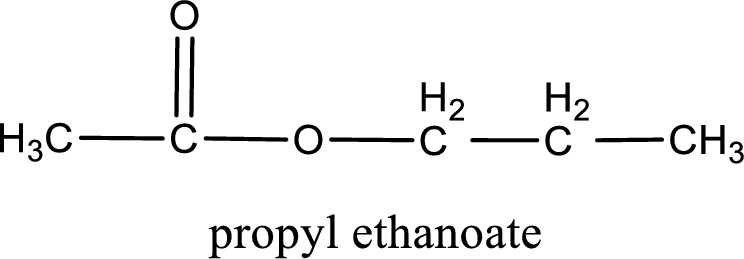
The reaction between two compounds that are shown above, result in the formation of ester. The structure of the ester formed and the complete reaction can be given as shown below,
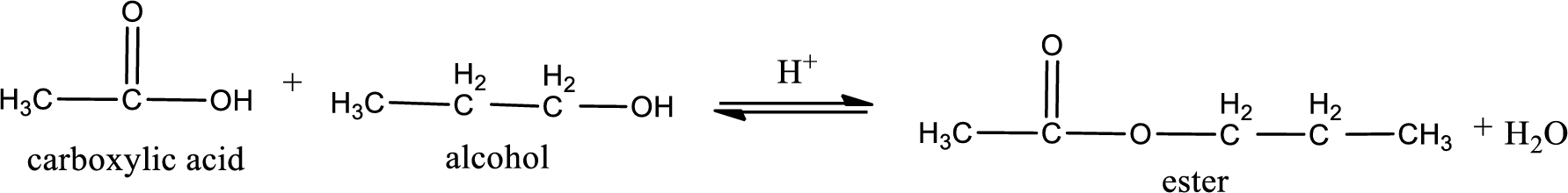
The structure of ester is,
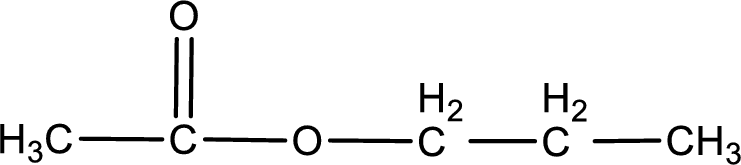
The alkyl and acyl group is identified as shown below,
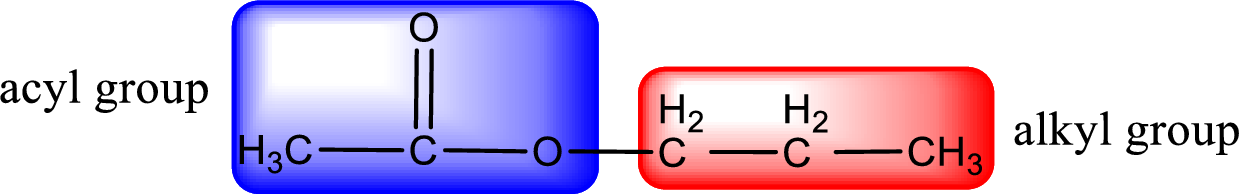
Alkyl group contains three carbon atoms. Hence alkyl group is named as propyl. The acyl group contains two carbon atoms. The IUPAC name of carboxylic acid that contains three carbon atoms is ethanoic acid. For naming acyl group in ester the suffix “-ic acid” is converted into “-ate”. This gives the name of acyl part as ethanoate. Therefore IUPAC name of the given ester is propyl ethanoate.
IUPAC name of the formed ester is assigned.
(b)
Interpretation:
The IUPAC name for the ester formed when acetic acid and 1-pentanol react has to be assigned.
Concept Introduction:
Esters are prepared by condensation of carboxylic acid with an alcohol. A molecule of water is lost on this reaction. The reaction that takes place in producing esters is known as esterification reaction.
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
For naming an ester, it can be structurally viewed in a way that contains an acyl group and an alkyl group.
Rules to obtain IUPAC name and common name for an ester:
- Alkyl part appears first in the IUPAC name and it is followed by the acyl part of ester as a separate word.
- Name of the alkyl part in the ester is just a name of R group. It can be alkyl, cycloalkyl, or aryl group.
- Acyl part present in the ester is named by considering the acid name and replacing the suffix “-ic acid” with “-ate”.
- To obtain the common name the alkyl part name is the same while the acyl part name is derived from the common name of the acid by replacing the suffix “-ic acid” with “-ate”.
(b)
Answer to Problem 16.98EP
IUPAC name of the ester formed is pentyl ethanoate.
Explanation of Solution
Given carboxylic acid and alcohol structure is,
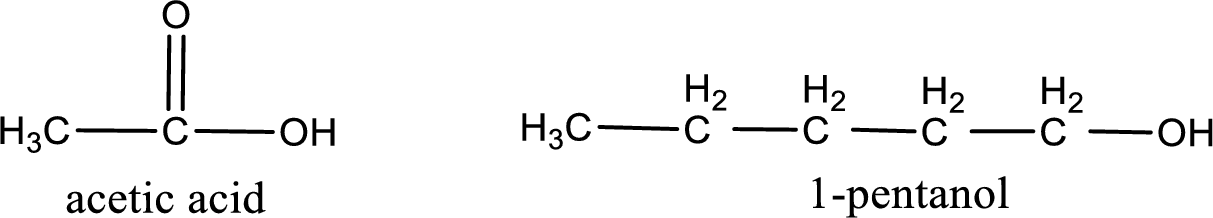
The reaction between two compounds that are shown above, result in the formation of ester. The structure of the ester formed and the complete reaction can be given as shown below,

The structure of ester is,
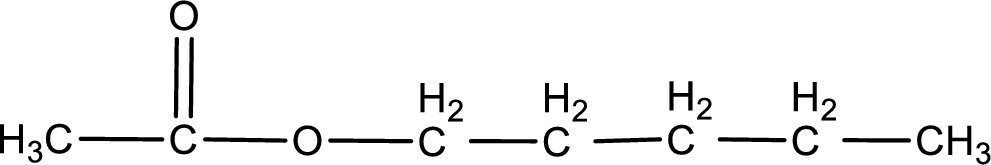
The alkyl and acyl group is identified as shown below,
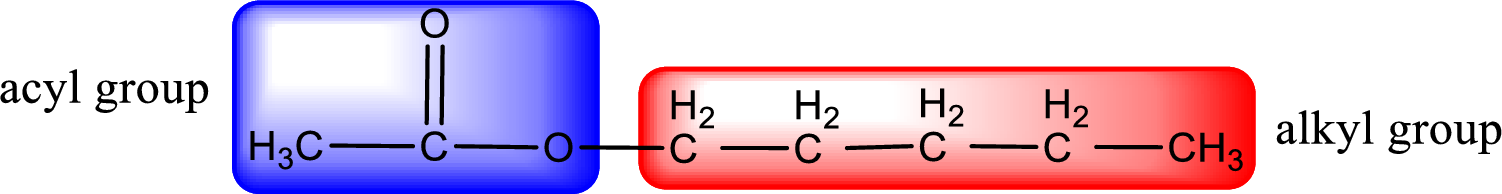
Alkyl group contains five carbon atoms. Hence alkyl group is named as pentyl. The acyl group contains two carbon atoms. The IUPAC name of carboxylic acid that contains three carbon atoms is ethanoic acid. For naming acyl group in ester the suffix “-ic acid” is converted into “-ate”. This gives the name of acyl part as ethanoate. Therefore IUPAC name of the given ester is pentyl ethanoate.
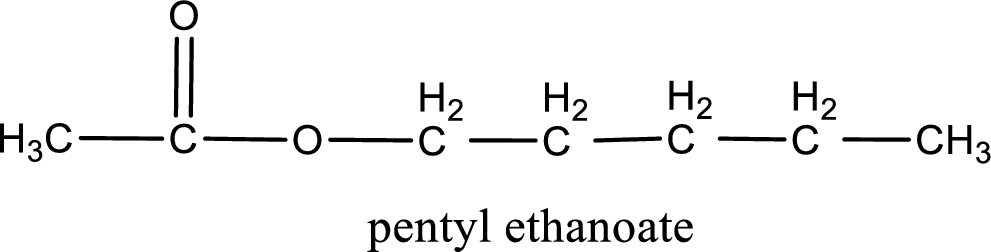
IUPAC name of the formed ester is assigned.
(c)
Interpretation:
The IUPAC name for the ester formed when acetic acid and 2-pentanol react has to be assigned.
Concept Introduction:
Esters are prepared by condensation of carboxylic acid with an alcohol. A molecule of water is lost on this reaction. The reaction that takes place in producing esters is known as esterification reaction.
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
For naming an ester, it can be structurally viewed in a way that contains an acyl group and an alkyl group.
Rules to obtain IUPAC name and common name for an ester:
- Alkyl part appears first in the IUPAC name and it is followed by the acyl part of ester as a separate word.
- Name of the alkyl part in the ester is just a name of R group. It can be alkyl, cycloalkyl, or aryl group.
- Acyl part present in the ester is named by considering the acid name and replacing the suffix “-ic acid” with “-ate”.
- To obtain the common name the alkyl part name is the same while the acyl part name is derived from the common name of the acid by replacing the suffix “-ic acid” with “-ate”.
(c)
Answer to Problem 16.98EP
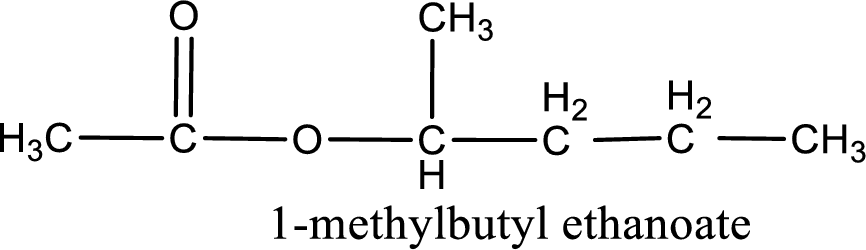
IUPAC name of the ester formed is 1-methylbutyl ethanoate.
Explanation of Solution
Given carboxylic acid and alcohol structure is,
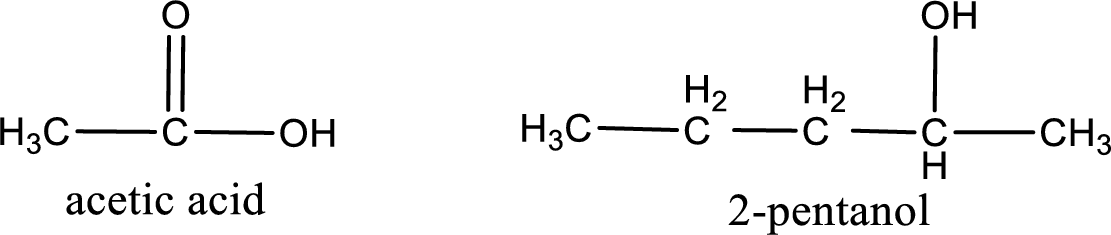
The reaction between two compounds that are shown above, result in the formation of ester. The structure of the ester formed and the complete reaction can be given as shown below,

The structure of ester is,
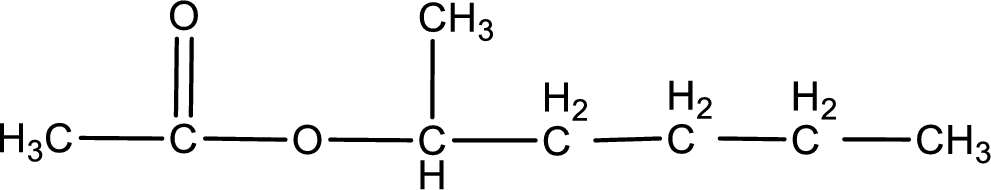
The alkyl and acyl group is identified as shown below,
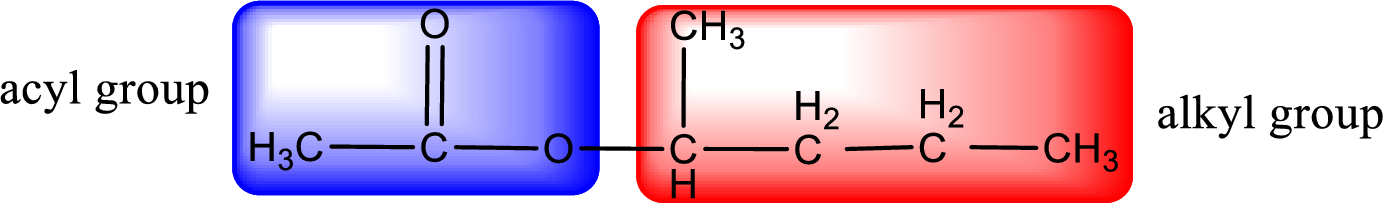
Alkyl group contains five carbon atoms. Four carbon in a long chain with a methyl group substituted in the first carbon atom. Hence alkyl group is named as 1-methylbutyl. The acyl group contains two carbon atoms. The IUPAC name of carboxylic acid that contains three carbon atoms is ethanoic acid. For naming acyl group in ester the suffix “-ic acid” is converted into “-ate”. This gives the name of acyl part as ethanoate. Therefore IUPAC name of the given ester is 1-methylbutyl ethanoate.
IUPAC name of the formed ester is assigned.
(d)
Interpretation:
The IUPAC name for the ester formed when ethanol and benzoic acid react has to be assigned.
Concept Introduction:
Esters are prepared by condensation of carboxylic acid with an alcohol. A molecule of water is lost on this reaction. The reaction that takes place in producing esters is known as esterification reaction.
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
For naming an ester, it can be structurally viewed in a way that contains an acyl group and an alkyl group.
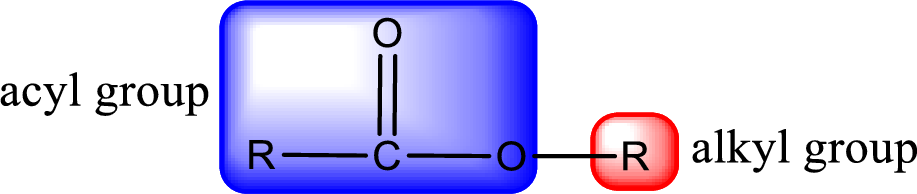
Rules to obtain IUPAC name and common name for an ester:
- Alkyl part appears first in the IUPAC name and it is followed by the acyl part of ester as a separate word.
- Name of the alkyl part in the ester is just a name of R group. It can be alkyl, cycloalkyl, or aryl group.
- Acyl part present in the ester is named by considering the acid name and replacing the suffix “-ic acid” with “-ate”.
- To obtain the common name the alkyl part name is the same while the acyl part name is derived from the common name of the acid by replacing the suffix “-ic acid” with “-ate”.
(d)
Answer to Problem 16.98EP
IUPAC name of the ester formed is ethyl benzoate.
Explanation of Solution
Given carboxylic acid and alcohol structure is,
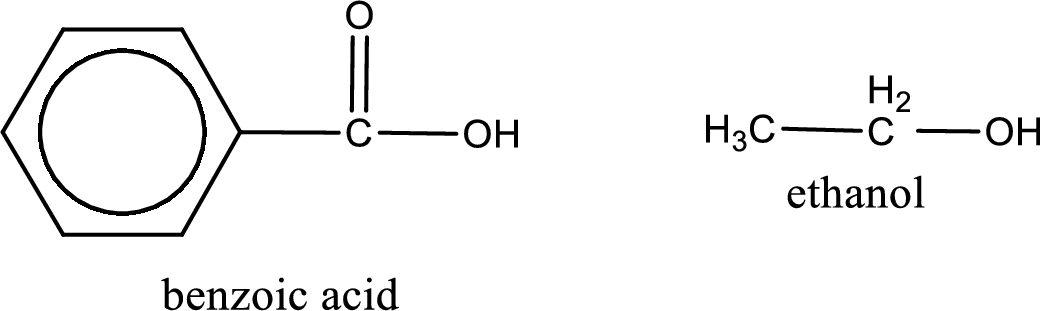
The reaction between two compounds that are shown above, result in the formation of ester. The structure of the ester formed and the complete reaction can be given as shown below,
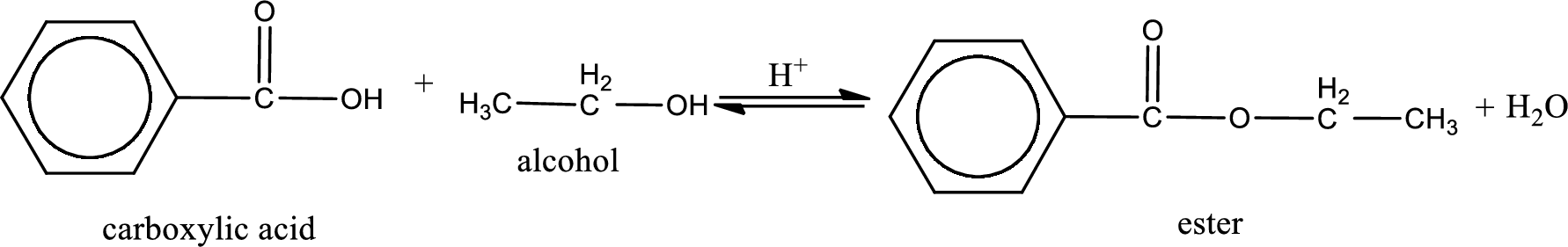
The structure of ester is,
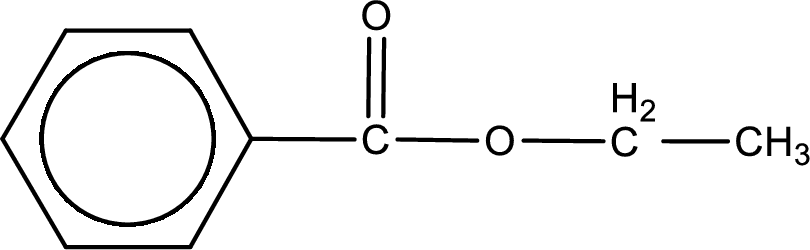
The alkyl and acyl group is identified as shown below,
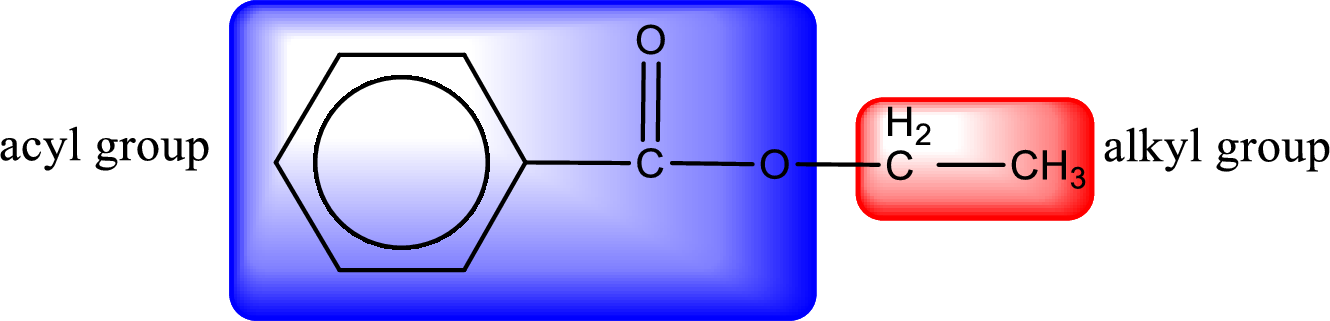
Alkyl group contains only two carbon atoms. Hence alkyl group is named as ethyl. The acyl group contains a benzene ring structure. The IUPAC name of carboxylic acid that contains benzene ring is benzoic acid. For naming acyl group in ester the suffix “-ic acid” is converted into “-ate”. This gives the name of acyl part as benzoate. Therefore IUPAC name of the given ester is ethyl benzoate.
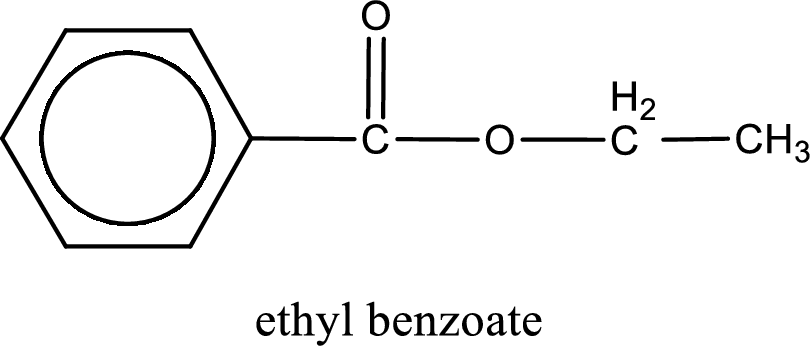
IUPAC name of the formed ester is assigned.
Want to see more full solutions like this?
Chapter 16 Solutions
General, Organic, and Biological Chemistry
- What are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. acetic acid and toluene b. phenylic acid and ethanol C. ethanoic acid and benzene d. benzoic acid and ethanol e. phenylic acid and methanol O a O barrow_forwardWhich of the following compounds can react with C2H5MgBr to give 3-pentanol? a.acetone b.ethanal c.acetic acid d.ethyl formatearrow_forwardDraw a structural formula for each of the following aldehydes. a. Acetaldehyde b. Butyraldehyde c. Dichloroacetaldehyde d. 2-Methylbenzaldehydearrow_forward
- 1. Draw the structural formula of the hemiacetal formed from each ơf the following pairs of reactants. a. Acetaldehyde and methanol 0 b. 2-Pentanone and ethyl alcohol С. Butanal and isopropyl alcohol d. Acetone and ethanolarrow_forwardEsters are formed by the reaction of carboxylic acids and acid in the presence of A. Alkanes B. Alcohols C. Alkenes D. Aldehydes E. Alkynesarrow_forwardDefine the structure of a carboxylic acid ?arrow_forward
- Write a general equation showing the preparation of a carboxylic acid from an alcohol.arrow_forwardExplain the characteristic reaction of aldehydes and ketones ?arrow_forward1. Which of the following structural features is possessed by aldehydes but not ketones? a. At least one hydroxyl group is bonded to the carbonyl carbon atom. b. At least one hydrogen atom is bonded to the carbonyl carbon atom. c. The carbonyl carbon atom is bonded to two other carbon atoms. d. The carbonyl carbon atom is part of a ring structure. 2. What is the IUPAC name of the following compound in the attached photo? a. 3-chloro-4-nitrobenzoic acid b. 2-chloro-4-carboxynitrobenzene c. 2-nitro-5-carboxychlorobenzene d. 2-chloro-1-nitro-4-benzoic acid 3. Which of the following is the correct bond-line structure for CH3C≡C(CH2)2CH(CH3)2? a. Structure I b. Structure II c. Structure III d. Structure IVarrow_forward
- For each of the esters provided, identify the alcohol and the carboxylic acid that reacted. a. methyl propanoate b. ethyl methanoatearrow_forwardA lot of controversy surrounds the use of Aspartame as an artificial sweetener. The main argument is concerned with the production of methanol in the body. From the structure of aspartame given below: G0₂CH3 HẠN CH—ỆNH—CHCH, CH, COCH Source for the formation of methanol is, A. ester group in the compound B. amide group in the compound C. carboxylic acid group in the compound D. amino group in the compound E. benzene ring in the compoundarrow_forwardWhich of the following compounds is solid at room temperature? O a. Acetic acid O b. Acetic acid O c. Linoleic acid O d. Benzoic acid e. Stearic acid Give the IUPAC of the compound, H₂N Br Answer: The product of the reaction between acetaldehyde and hydrogen cyanide is generally known as a. Ester O b. Grignard reagent O c. Cyanohydrin O d. Nitrile e. Alcoholarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



