
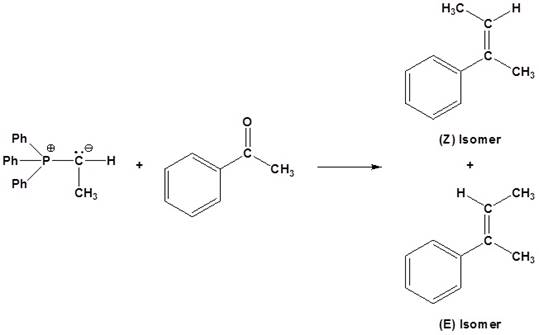
(a)
Interpretation:
The set of reagents satisfying the given conditions used for the synthesis of the given
Concept introduction:
An
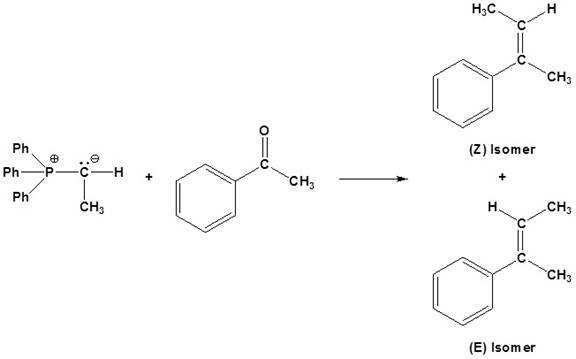
(b)
Interpretation:
The
Concept introduction:
An aldehyde or a ketone reacts with phosphonium ylide to form an alkene. This reaction is known as wittig reaction.
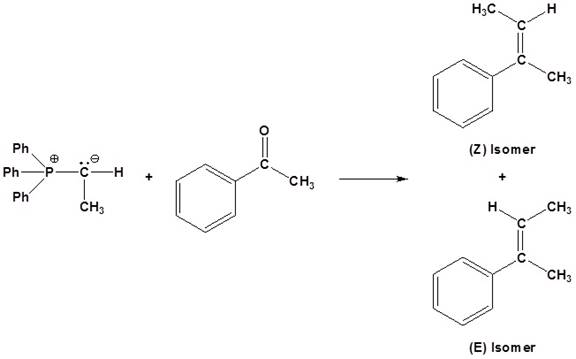
(c)
Interpretation:
The reagents used for the synthesis of given set of alkenes should be determined.
Concept introduction:
An aldehyde or a ketone reacts with phosphonium ylide to form an alkene. This reaction is known as wittig reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry (8th Edition)
- a. HO b. NH 33. Which is a better nucleophile? a. H₂O or HO c. H₂S b. NH3 or NH₂ C. CH, CO in a better leaving group. or CH3CH₂O d. -0 or -0arrow_forwardConsider the following nucleophilc substitution. LOCH3 Br OCH3 в A a. Identify the reaction conditions A and specify the reaction mechanism for the nucleophilic substitution. Explain your choice. b. Explain why the transformation does not give rise to significant elimination. c. Which product(s) B is formed in the reaction? Please specify the stereochemistry clearly with reference to the reaction mechanism.arrow_forwardWhich of the following compounds cannot be prepared by a Heck reaction? a. For those compounds that can be prepared by a Heck reaction, what starting materials are required?arrow_forward
- Enols are quite reactive toward electrophiles than alkenes because: а. The OH group has a powerful electron-donating O resonance effect b. A resonance structure can be drawn that places a negative charge on one of the carbon atoms, making this carbon electrophilic С. The enol can react with nucleophilic carbon to form a new bond to carbon d. None of the options are correctarrow_forward12. What alkyl halide and nucleophile are needed to prepare the following compounds? a. b. OHarrow_forwarda. Which of the following compounds cannot be prepared by a Heck reaction?b. For those compounds that can be prepared by a Heck reaction, what starting materials are required?arrow_forward
- Devise a synthesis of each compound using CH3CH₂CH=CH₂ as the starting material. You may use any other organic compounds or inorganic reagents. a. b. d. e. Br Br Br OH OH (+ enantiomer)arrow_forwardShow how to prepare each Gilman reagent from an appropriate alkyl or vinylic halide. Q. 3-Methyloctanearrow_forwardThe major product that would form from the presented reaction scheme is? .C. CH3 H2/Pd NO2arrow_forward
- Select all compounds capable of keto-enol tautomerism. он H.C. он H.C. H.C. H.C. CH, CH, CH, H.C H.C a b darrow_forward4. Propose a synthesis of each of the following compounds using the indicated starting material. You may use any organic compounds and any inorganic compounds or solvents of your choice. Do not show any reactive intermediates, mechanisms, or transition states, but be sure to show each isolable compound along your synthetic route. a. b. C. H3 -CH3 steps Ph Ph steps 4 CN steps -CH₂ Ph Ph -OH (racemic)arrow_forward4. Provide a synthesis for each of the molecules shown starting from benzene and any other reagents necessary. a.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





