
(a)
Interpretation:
A mechanism of given reaction under neutral condition is to be drawn.
Concept introduction:
The carbonyl carbon is electrophilic in nature due to the electronegativity difference between the carbonyl carbon and oxygen. Since water is amphoteric in nature can act as acid as well as a base depending on availability and the reaction condition. Under neutral conditions, water acts as the nucleophile.
Answer to Problem 18.1P
A mechanism of given reaction under neutral condition is:
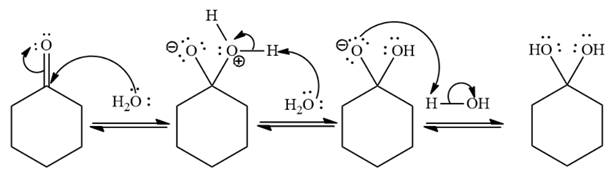
Explanation of Solution
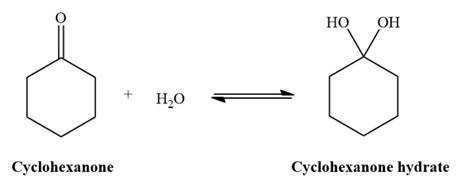
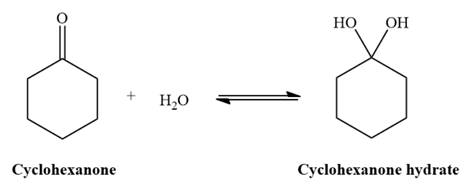
The given reaction is:
Under neutral conditions, water acts as the nucleophile. The electrophilic addition step increases the total number of charges by two as shown below,
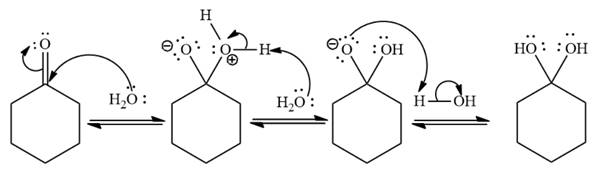
Under neutral conditions, water acts as a nucleophile.
(b)
Interpretation:
A mechanism of given reaction under basic condition is to be drawn.
Concept introduction:
Under basic conditions, the nucleophile becomes stronger and the electrostatic attraction is increased when the uncharged nucleophile is converted into a negatively charged nucleophile.
Answer to Problem 18.1P
A mechanism of given reaction under neutral condition is:
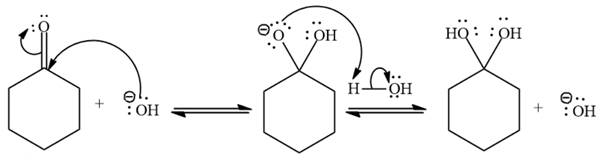
Explanation of Solution
The given reaction is:
Under basic conditions,
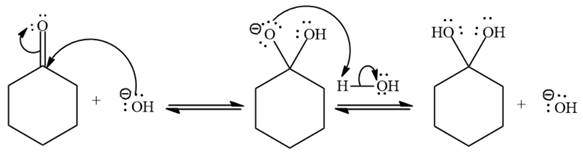
Under basic conditions, the nucleophile becomes stronger.
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





