
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 18, Problem 32P
Interpretation Introduction
Interpretation:
Compound that can remove
Concept introduction:
- Catalyst: Substance which helps in increasing the rate of a particular reaction without getting consumed in the reaction.
- Base Catalyst: A catalyst which helps in increasing the rate of a particular reaction by the removal of a proton. There are two types of catalysis:
- 1. Specific-base catalysis: Proton is completely removed before the slow step in a reaction
- 2. General- base catalysis: Proton is completely removed during the slow step in a reaction
- Intramolecular general-base catalysis: A catalysis where catalyst is a part of the molecule that undergoes the reaction promotes the reaction in a species by completely removing a proton during the slow step in a reaction
Expert Solution & Answer
Explanation of Solution
In the below compound, negatively charged oxygen atom is in axial position where it can easily undergo intramolecular general-base reaction comparing to
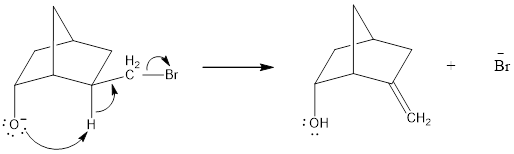
Therefore, the compound where
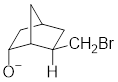
Want to see more full solutions like this?
Subscribe now to access step-by-step solutions to millions of textbook problems written by subject matter experts!
Students have asked these similar questions
Which of the following compounds is likely to undergo a gas-evolution reaction in an aqueous solution?
H2SO4
CH3COOH
H2CO3
HF
HCl
102.
The major products A and B in the following reaction sequence are
CHO
Br
Br
(i) PPh3
(ii) NaH
(A)
NaH
OH
(B)
Which of the following is the correct step of protonation of tran-2-butene?
A
B
C
D
Chapter 18 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 18.1 - Prob. 1PCh. 18.2 - If H218O were used to hydrolyze lysozyme, which...Ch. 18.3 - Which of the following amino acid side chains can...Ch. 18.3 - Arginine and lysine side chains fit into trypsins...Ch. 18.4 - Which of the following amino acid side chains can...Ch. 18.4 - Prob. 6PCh. 18.5 - Prob. 7PCh. 18.5 - Draw the mechanism for the hydroxide-ion-catalyzed...Ch. 18.5 - What advantage does the enzyme gain by forming an...Ch. 18.7 - Prob. 10P
Ch. 18.7 - Prob. 11PCh. 18.8 - How many conjugated double bonds are there in a....Ch. 18.8 - Instead of adding to the 4a-position and...Ch. 18.8 - In succinate dehydrogenase, FAD is covalently...Ch. 18.8 - Prob. 15PCh. 18.9 - Acetolactate synthase is another TPP-requiring...Ch. 18.9 - Acetolactate synthase can also transfer the acyl...Ch. 18.9 - Prob. 18PCh. 18.9 - Prob. 19PCh. 18.10 - Prob. 21PCh. 18.11 - Prob. 23PCh. 18.11 - Which compound is more easily decarboxylated?Ch. 18.11 - Explain why the ability of PLP to catalyze an...Ch. 18.11 - Explain why the ability of PLP to catalyze an...Ch. 18.12 - What groups are interchanged in the following...Ch. 18.13 - Why is the coenzyme called tetrahydrofolate?Ch. 18.13 - What amino acid is formed by the following...Ch. 18.13 - How do the structures of tetrahydrofolate and...Ch. 18.13 - What is the source of the methyl group in...Ch. 18 - Prob. 32PCh. 18 - Prob. 33PCh. 18 - From what vitamins are the following coenzymes...Ch. 18 - Prob. 35PCh. 18 - For each of the following reaction, name both the...Ch. 18 - Explain why serine proteases do not catalyze...Ch. 18 - Prob. 38PCh. 18 - For each of the following enzyme catalyzed...Ch. 18 - Trisephosphate isomerase (TIM) catalyzes the...Ch. 18 - Prob. 41PCh. 18 - What acyl groups have we seen transferred by...Ch. 18 - When UMP is dissolved in T2O, exchange of T for H...Ch. 18 - Prob. 44PCh. 18 - When transaminated, the three branched-chain amino...Ch. 18 - Aldolase shows no activity if it is incubated with...
Knowledge Booster
Similar questions
- Please show the arrow pushing for the following reaction. How would this be different if it was in basic conditions? ee H H+arrow_forwardIdentify the set of reaction conditions that best facilitates this reaction. Select the single best answer.arrow_forwardHow many different elimination products could be formed in the given molecule? (Hint: include (E) and (Z) isomers) O O A B 3 4 C 5 D 6 Brarrow_forward
- 6. Complete reaction scheme below indicating reagents, catalysts and conditions, and side product trigil bezinslog si6101fon 290b 1l (b Senines levirba 6 gaubong nasamots gniwollot ads to doinW 01 sniowl (6 CH3 40 CH3 Scansgowi (d Ege nodie) ( nsgyxo (b NO₂ 19m02 2i gniwollet sits to mainW II HOarrow_forward5- Does SN2 occur in one step while SN1 occurs in three steps?arrow_forwardThe reaction of the carbonyl compound with cyanide yields The R-isomer The S-isomer Both the R and S isomer None of the above Adding NaCN to the carbonyl compound poses the risk of making HCN, which is poisonous. How can this risk be mitigated? Add NaOH first, then NaCN Add NaCN first, then NaOH Add NaHSO3 first, then NaCN Add NaOH first, then NaHSO3arrow_forward
- 3-If the p-value of alkaline saponification of methylbenzoate is 2.38 and the rate constant for saponification of methylbenzoate under these conditions is 2x104 calculate the rate constant for hydrolysis of methyl-m-nitrobenzoate.arrow_forwardWhich of the following Bronsted-Lowry conjugate pairs is/are CORRECT? H3PO4 – PO43- C6H5COOH – C6H5COO- CH3CH2NH3+ – CH3CH2NH2 Which of the following Bronsted-Lowry conjugate pairs is/are CORRECT? H3PO4 – PO43- C6H5COOH – C6H5COO- CH3CH2NH3+ – CH3CH2NH2 II only I only I and III II and IIIarrow_forward7. Reaction Scheme. NH₂ NH2 or two differnet methods (no same steps/reagents) C5H12N2 1. xs Mel, xs K2CO3 2. Ag2O, H₂O 3. heat Br2, xs NaOH, xs H₂O OHC 1.03 2. DMS CHOarrow_forward
- Which of the following species are not nucleophiles: Na+, Cl-, +CH3, :NH3, HO:-?arrow_forwardWhich of the following reactions will be fastest? (a) Br: (c) Br: H S. CF3 (b) Br: (d) Br: CH3 OHarrow_forwardowing reactions proceeds via an SN1 or SN2 of the reaction: (b) S2 pro85 39 Br tort atubong auonsV HMPAnoitutitaduaarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT