
Identify the elements used in each example of molecular art.

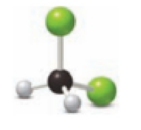
(a)
Interpretation:
The element in the molecular art has to be given.
The molecular art is,

Figure 1
Explanation of Solution
The red spheres in the above space filling figure 1 represents oxygen and the black spheres represent the carbon molecule. Thus, one particle contains two oxygen molecule and one particle contains one oxygen and one carbon molecule and another particle contains two oxygen and one carbon molecule.
(b)
Interpretation:
The element in the molecular art has to be given.
The molecular art is,
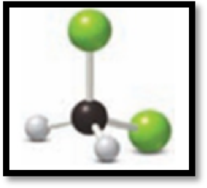
Figure 2
Explanation of Solution
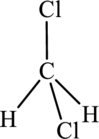
This ball and stick model represents dichloromethane. The molecule contains two Chlorine molecules (green balls), one carbon (black ball) and two hydrogen molecule (grey balls).
The structure of dichloromethane is,
Want to see more full solutions like this?
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
- Name and give symbols for three transition metals in the fourth period. Look up each of your choices in a dictionary, a book such as The Handbook of Chemistry and Physics, or on the Internet, and make a list of their properties. Also list the uses of each element.arrow_forwardA material is believed to be a compound. Suppose you have several samples of this material obtained from various places around the world. Comment on what you would expect to find upon observing the melting point and color for each sample. What would you expect to find upon determining the elemental composition for each sample?arrow_forwardClassify each of these as an element, a compound, a heterogeneous mixture, or a homogeneous mixture. Explain your choice in each case. (a) Chunky peanut butter (b) Distilled water (c) Platinum (d) Airarrow_forward
- A materials engineer has filed for a patent for a new alloy to be used in golf club heads. The composition by mass ranges from 25 to 31% manganese, 6.3 to 7.8% aluminum, 0.65 to 0.85% carbon, and 5.5 to 9.0% chromium, with the remainder being iron. What are the maximum and minimum percentages of iron possible in this alloy? Use Figure 2.12 to snake a prediction about how the density of this alloy would compare with that of iron; justify your prediction.arrow_forwardWhich part of the description of a compound or element refers to its physical properties and which to its chemical properties? (a) The colorless liquid ethanol bums in air. (b) The shiny metal aluminum reacts readily with orange-red brominearrow_forwardClassify each of the following observations about the behavior of a substance as a physical property or chemical property of the substance. a. does not tarnish when exposed to dry air at 25C b. does tarnish when exposed to moist air at 25C c. is a liquid at 75C d. is a solid at 15Carrow_forward
- Please use this word bank to fill in the blanks of the first 5 questions. Atom 1. S is a/an 2. Sg is a/an (Yes! You can use more than one per blank spot!) element molecule Homogenous mixture 3. MgSO4 is a/an 4. A sample of filtered seawater containing Li+, Na+, K+, SO42, NO3¹, and Cl is a/an 5. A grey sample of basalt riddled with golden cubes of pyrite (FeS₂) is a/an 6. What is the state of S8 (s) at room temperature? compound Heterogeneous mixture 7. Give the full atomic symbol of S. 8. Give the subatomic particles of S.arrow_forwardList the main characteristics that an Earth material must possess to be considered a mineral and describe each. Compare and contrast the three primary particles contained in atoms. Distinguish among ionic bonds, covalent bonds, and metallic bonds.arrow_forwardThere are four sketches below. The first sketch shows a sample of Substance X. The three sketches underneath it show three different changes to the sample. You must decide whether each of these changes is possible. If a change is possible, you must also decide whether it is a physical change or a chemical change. Each sketch is drawn as if the sample were under a microscope so powerful that individual atoms could be seen. Also, you should assume that you can see the entire sample, and that the sample is in a sealed box, so that no matter can enter or leave. Change 1 Change 1 is: 00 impossible a physical change a chemical change Sample of Substance X 000 Change 2 Change 2 is: impossible a physical change a chemical change Change 3 Change 3 is: 00 impossible a physical change a chemical changearrow_forward
- define, compare and contrast what are meant by the emperical and molecular formulas for a substance . Give an example of eacharrow_forwardwhat is organic,inorganic, and physical chemistry? give 10 differences with your own wordsarrow_forwardThere are four sketches below. The first sketch shows a sample of Substance X. The three sketches underneath it show three different changes to the sample. You must decide whether each of these changes is possible. If a change is possible, you must also decide whether it is a physical change or a chemical change. Each sketch is drawn as if the sample were under a microscope so powerful that individual atoms could be seen. Also, you should assume that you can see the entire sample, and that the sample is in a sealed box, so that no matter can enter or leave. Sample of Substance X Change 1 Change 1 is O impossible O O a chemical change a physical change Change 2 Change 2 is: impossible O a physical change O a chemical change Change 3 000 000 Change 3 is: O impossible O a physical change O a chemical change Xarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





