
(a)
Interpretation:
The product formed when butyric acid reacts with ethanol in the presence of
Concept introduction:
Esterification occurs when a
Answer to Problem 20.27AP
The product formed when butyric acid reacts with ethanol in the presence of
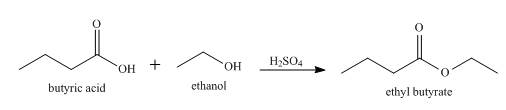
Explanation of Solution
When butyric acid reacts with ethanol in the presence of

Figure 1
When butyric acid reacts with ethanol in presence of
(b)
Interpretation:
The product formed when butyric acid reacts with aqueous
Concept introduction:
An acid-base reaction occurs through a transfer of proton from an acid to a base. The base gets protonated and the acid gets deprotonated. The reaction results in the formation of a conjugate acid and a conjugate base as the products. The type of products formed affects the direction of reaction at equilibrium.
Answer to Problem 20.27AP
The product formed when butyric acid reacts with aqueous
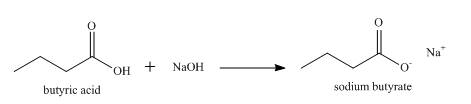
Explanation of Solution
When a carboxylic acid reacts with a base, the hydrogen atom is abstracted by the base and a salt of carboxylic acid is formed.
So, when butyric acid reacts with aqueous

Figure 2
The product formed when butyric acid reacts with aqueous
(c)
Interpretation:
The product formed when butyric acid reacts with
Concept introduction:
The substances which on addition removes oxygen atom or hydrogen atom from the other substance, that is, reduces the other substances are known as reducing agents. Reducing agents themselves get oxidized. Carboxylic acid is reduced when reacts with reducing agent,
Answer to Problem 20.27AP
The product formed when butyric acid reacts with
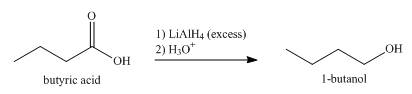
Explanation of Solution
Carboxylic acid is reduced when reacts with reducing agent,
When butyric acid reacts with excess of

Figure 3
When butyric acid reacts with excess of
(d)
Interpretation:
The product formed when butyric acid is heated is to be stated.
Concept introduction:
Carboxylic acids such as
Answer to Problem 20.27AP
When butyric acid is heated, no product is formed.
Explanation of Solution
The structure of butyric acid does not have any electron-withdrawing group like
Butyric acid on heating gives no reaction.
(e)
Interpretation:
The product formed when butyric acid reacts with
Concept introduction:
Carboxylic acids react with
Answer to Problem 20.27AP
The product formed when butyric acid reacts with
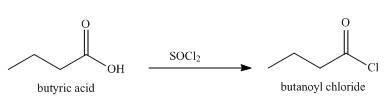
Explanation of Solution
When butyric acid reacts with

Figure 4
The product formed when butyric acid reacts with
(f)
Interpretation:
The product formed when butyric acid reacts with diazomethane in ether is to be stated.
Concept introduction:
Esterification occurs when carboxylic acid reacts with diazomethane in ether. Esters end with a suffix,
Answer to Problem 20.27AP
The product formed when butyric acid reacts with diazomethane in ether is shown below.
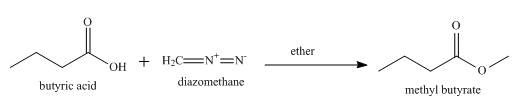
Explanation of Solution
Esterification occurs when butyric acid reacts with diazomethane in ether and methyl butyrate is formed as a product along with the release of nitrogen gas. The complete reaction is shown below.

Figure 5
The product formed when butyric acid reacts with diazomethane in ether is methyl butyrate.
(g)
Interpretation:
The product formed when
Concept introduction:
Acetal is formed when an alcohol in excess reacts with an aldehyde or ketone in acidic medium. The
Answer to Problem 20.27AP
The product formed when
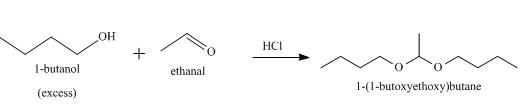
Explanation of Solution
When an alcohol reacts with an aldehyde in presence of an acid catalyst, a hemiacetal is formed as an intermediate which further reacts to produce acetal with the release of water. The acidic catalyst is used because the alcohol is a weak nucleophile. So on addition of an acid, the oxygen of aldehyde gets protonated and a carbocation is formed. The alcohol easily attacks the carbocation to form the germinal diether derivative of aldehyde, that is, an acetal. When

Figure 6
When
(h)
Interpretation:
The product formed when butanoyl chloride reacts with benzene in
Concept introduction:
Friedel crafts acylation reaction is a reaction in which an
Answer to Problem 20.27AP
The product formed when butanoyl chloride reacts with benzene in
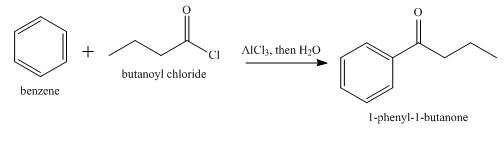
Explanation of Solution
Benzene undergoes Friedel Crafts acylation reaction on reaction with butanoyl chloride in presence of

Figure 7
The product formed when butanoyl chloride reacts with benzene in
(i)
Interpretation:
The product formed when
Concept introduction:
Wolff Kishner Reduction is a reaction in which aldehydes and ketones are converted into
Answer to Problem 20.27AP
The product formed when
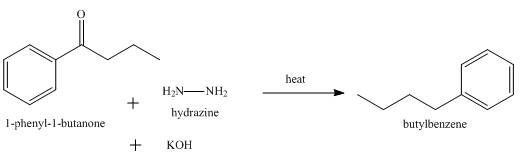
Explanation of Solution
Wolff Kishner reduction takes place when

Figure 8
The product formed when
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry
- provide the structure of the intermediate and product for the following reaction : (c) H CH,OH/H (C)arrow_forwardWhen the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardThe odor of ripe bananas and many other fruits is due to the presence of esters. For example: Banana oil (isopentyl acetate) (a) Write the name (common or IUPAC) of the ester responsible for the fragrance of the following: pineapple, orange, apple, peach, & lavender (b) Choose one fragrant from (a) and name the alcohol and the carboxylic acid needed to synthesize this ester. (c) Show the detailed mechanism of the Fischer Esterification reaction that will be involved in the synthesis of the fragrant you have chosen in part (a).arrow_forward
- Identify (A) in the following reaction. 2H2 Pt (A) KMNO4 Warm conc. || С — С — о—н |CO,H + HO CO2H cis-cyclo hexane 1,2-dicarboxylic acid (a) (b) (c) (d)arrow_forwardDimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of female hamsters, acts as a sexual attractant for the male hamster. Write an equation for its synthesis from methanethiol.arrow_forward(i) State reagents G and J. (ii) Draw the structural formula for compounds D, E and H.arrow_forward
- Arrange these compounds in order of increasing boiling point (values in °C are -42, -24, 78, and 118). (a) CH,CH,OH (b) CH,OCH, (c) CH,CH,CH, (d) CH,COOHarrow_forwardPredict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.(a) PhMgBr, then H3O+ (b) Tollens reagent (c) semicarbazide and weak acid(d) excess ethanol and acid (e) propane-1,3-diol, H+ (f) zinc amalgam and dilute hydrochloric acidarrow_forwardCH₂-CH₂-CH₂-C-OH butanoic acid CH₂ HO–C–CH,CHCH,—CHO 3-methylpentanoic acid Name each carboxylic acid. (a) HO CH₂ CH₂ 99 - 0 B) CHICHICIO CH, Draw a condensed structural diagram for each carboxylic acid (a) Hexanoic acid (b) 3-propyloctanoic acid Iarrow_forward
- Give reasons for the following :(i) Phenol is more acidic than methanol.(ii) The C—O—H bond angle in alcohols is slightly less than the tetrahedral angle (190°28′).(iii) (CH3)3C—O—CH3 on reaction with HI gives (CH3)3C—I and CH3—OH as the main products and not (CH3)3C—OH and CH3—I.arrow_forwardBent (D) Trigonal pyramidal 5. A student wishes to prepare ethyl acetate from the reaction of ethanol and acetic acid. To be successful, this reaction requires (A) an acidic catalyst. (C) an oxidizing agent. (B) a basic catalyst. (D) a reducing agent. 6. Which alkyl halide reacts most rapidly with aqueous sodium hydroxide solution? (A) CH₂Cl (B) CH₂I (C) (CH3)3CCH₂Cl (D) (CH3)3CCH₂I 57. How many isomers are there with the formula C6H₁4?arrow_forwardthe fragrant ester benzyl acetate is a naturally occurring compound in many flowers. The flavor and fragrance industry produces this compound by a reaction of benzyl alcohol with excess acetic acid, using hydrochloric acid as a catalyst. Water is a co-product of the reaction. Provide the reaction scheme.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
