
Concept explainers
Draw the product when each compound is treated with either
a. 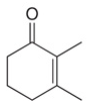 b.
b. 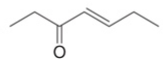 c.
c. 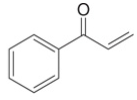
(a)
Interpretation: The product formed by the treatment of given compound with either
Concept introduction: Organometallic reagents like
Answer to Problem 20.34P
The product formed by the treatment of given compound with
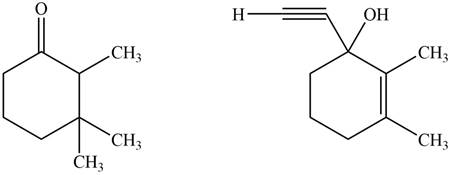
Figure 1
Explanation of Solution
The given reagents are organocuprate
The product formed by the reaction of
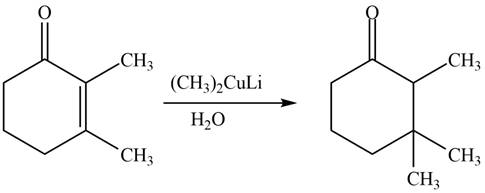
Figure 2
The product formed by the reaction of
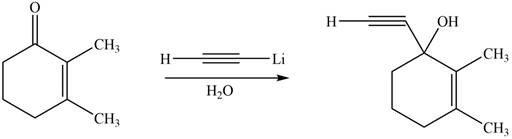
Figure 3
The product formed by the treatment of given compound with
(b)
Interpretation: The product formed by the treatment of given compound with either
Concept introduction: Organometallic reagents like
Answer to Problem 20.34P
The product formed by the treatment of given compound with
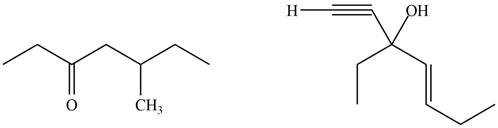
Figure 4
Explanation of Solution
The given reagents are organocuprate
The product formed by the reaction of
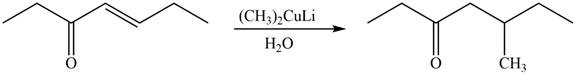
Figure 5
The product formed by the reaction of
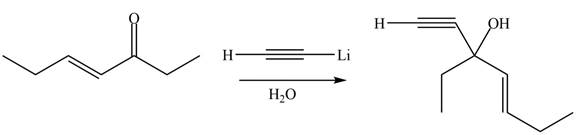
Figure 6
The product formed by the treatment of given compound with
(c)
Interpretation: The product formed by the treatment of given compound with either
Concept introduction: Organometallic reagents like
Answer to Problem 20.34P
The product formed by the treatment of given compound with
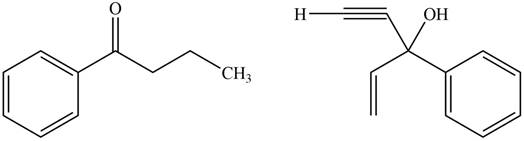
Figure 7
Explanation of Solution
The given reagents are organocuprate
The product formed by the reaction of
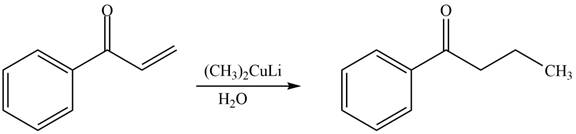
Figure 8
The product formed by the reaction of
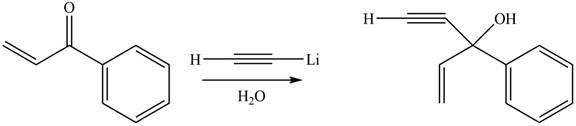
Figure 9
The product formed by the treatment of given compound with
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry
- Show how to bring about each conversion in good yield. a. b. C6H5 Cl OH COOH C6H5 COOHarrow_forwardCompounds that contain both a hydroxyl group (OH) and a carboxyl group (COOH) can undergo an intramolecular esterifi cation reaction. What product is formed when each hydroxy acid undergoes an intramolecular reaction? a. HOCH 2CH 2CH 2CH 2CO 2H b. HOCH 2CH 2CH 2CO 2Harrow_forwardDraw the product formed when A is treated with each series of reagents.a. [1] H2O; [2] NaH; [3] CH3Brb. [1] CuCN; [2] DIBAL-H; [3] H2O c. [1] C6H5NH2; [2] CH3COClarrow_forward
- Draw the products formed when each compound is treated with CH;CH,COCI, AICI3. CH(CH3)2 N(CH)2 Br CH3 CH(CH)2 a. b. C. d. е.arrow_forwardConvert each compound to its enol or keto tautomer. HO- a. b. CH CH3arrow_forwardAnswer each question for A and B depicted in the ball-and-stick models.a. What is the IUPAC name for each compound?b. What product is formed when each compound is treated with NaOH?c. Name the products formed in part (b).d. Draw the structure of an isomer that is at least 105 times less acidic than each compound.arrow_forward
- Draw the products formed when each compound is treated with HNO3 and H2SO4.State whether the reaction occurs faster or slower than a similar reaction with benzene.arrow_forwardDraw a stepwise mechanism for the following reaction. CH,O OCH, H2SO, H20 2 CH;OHarrow_forwardSynthesize each compound from acetylene. You may use any other organic or inorganic reagents. C. CH,CH,CH,CH, CHO d. CH3CH, CHỖ CHarrow_forward
- Draw the stepwise mechanism for the following reaction. CH;CH2OH H2SO4arrow_forwardWhich is a thiol? CH3 HS SH b. HO. CH3 H.CS CHa CH C. H. d. భిగింగడి 工 a.arrow_forwardDraw the products formed when phenol (C6H5OH) is treated with each set of reagents.a. [1] HNO3, H2SO4; [2] Sn, HCIb. [1] (CH3CH2)2CHCOCI, AlCl3; [2] Zn(Hg), HCIc. [1] CH3CH2CI, AlCl3; [2] Br2, hvd. [1] (CH3)2CHCI, AlCl3; [2] KMnO4arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





