
Figure 20-6 is the
(a)
The temperature of the gas at B, if the temperature at A is
Answer to Problem 52SP
Solution:
Explanation of Solution
Given data:
Refer to Fig. 20-6.
The temperature at A is
The mass of the ideal gas enclosed is
The specific heat of the gas at constant volume is
The gas follows the process A to B in the thermodynamic cycle shown in Fig. 20-6.
Formula used:
The gas equation between initial and final condition of a gas can be written as,
Here,
The formula for the conversion of the initial temperature of a gas from the Celsius scale to the Kelvin scale is,
Here,
The area of a trapezium is calculated by the formula,
Here,
The work done in a thermodynamic process is given by the area under the line representing the process in the pressure-volume diagram.
Here,
Explanation:
Draw the thermodynamic cycle diagram given in Fig- 20.6.
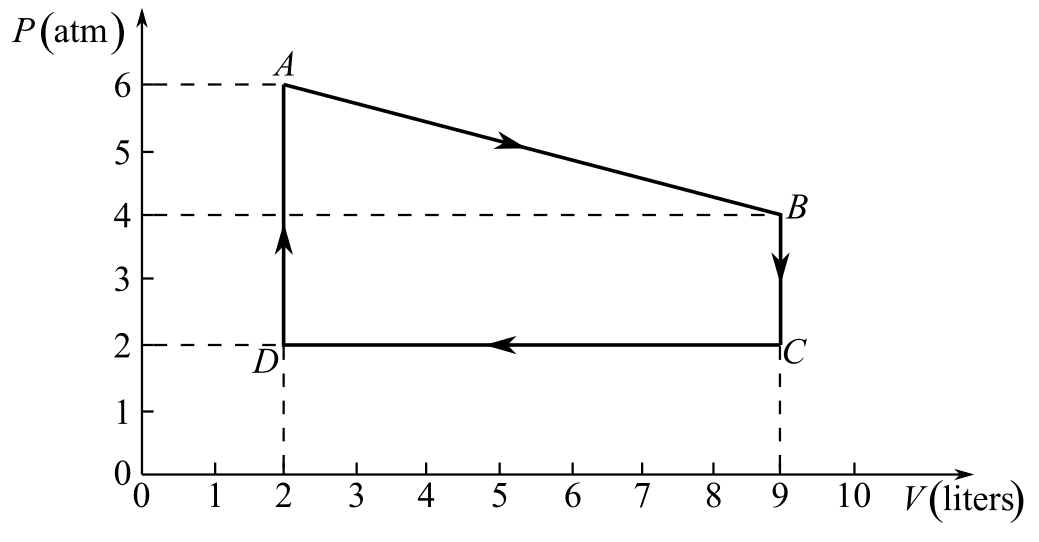
Recall the expression for the conversion of temperature at A from Celsius to Kelvin.
Here,
Substitute
Refer to the diagram and write the values of pressure and volume at points A and B, respectively.
And,
Here,
Recall the gas equation between points A and B.
Here,
Substitute
Conclusion:
The temperature at point B is
(b)
The value of
Answer to Problem 52SP
Solution:
Explanation of Solution
Given Data:
Refer to Fig. 20-6.
The temperature at A is
The mass of the ideal gas enclosed is
The specific heat of the gas at constant volume is
Formula Used:
The formula for change in internal energy is,
Here,
The formula for conversion of temperature of gas from Kelvin scale to Celsius scale is,
Here,
Explanation:
Draw the thermodynamic cycle diagram given in Fig- 20.6.

Calculate the temperature at point B in Celsius.
Here,
Substitute
Calculate the change in temperature from A to B.
Substitute
Recall the formula for change in internal energy.
Substitute
Conclusion:
The value of
(c)
The value of
Answer to Problem 52SP
Solution:
Explanation of Solution
Given Data:
Refer to Fig. 20-6.
The temperature at A is
The mass of the ideal gas enclosed is
The specific heat of the gas at constant volume is
Formula Used:
The area of a trapezium is calculated by the formula,
Here,
The work done in a thermodynamic process is given by the area under the line representing the process in the pressure-volume diagram.
Here,
Explanation:
Draw the thermodynamic cycle diagram given in Fig- 20.6.

Understand that the work done in the thermodynamic process AB is equal to the area of the pressure-volume diagram under the line AB.
Draw the thermodynamic cycle diagram showing the area under the line AB.
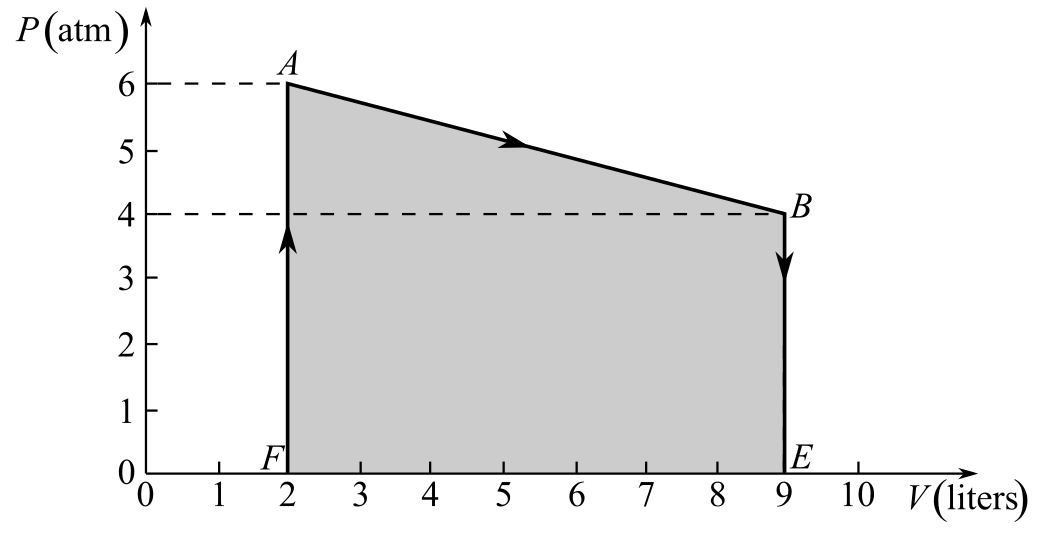
Here, the points E and F are as shown in the figure and the work done during the process AB is represented by the area under the line AB, which is equal to the area of the trapezium ABEF.
Refer to the figure and write the values of the lengths of sides AF, BE, and EF.
Recall the expression for the area of trapezium ABEF to calculate the area under the line AB in order to calculate the work done in the process AB.
Here,
Substitute
Recall the expression for the net-work done in a thermodynamic process in terms of the area of the pressure-volume diagram.
Substitute
The work done from A to B is
Conclusion:
The value of
(d)
The value of
Answer to Problem 52SP
Solution:
Explanation of Solution
Given Data:
Refer to Fig. 20-6.
The temperature at A is
The mass of the ideal gas enclosed is
The specific heat of the gas at constant volume is
Formula Used:
The first law of thermodynamics for a process is written as,
Here,
Explanation:
Draw the thermodynamic cycle diagram given in Fig- 20.6.

Recall the expression for the first law of thermodynamics for the process AB.
Substitute
Conclusion:
The value of
Want to see more full solutions like this?
Chapter 20 Solutions
Schaum's Outline of College Physics, Twelfth Edition (Schaum's Outlines)
- On a hot summer day, the density of air at atmospheric pressure at 35.0C is 1.1455 kg/m3. a. What is the number of moles contained in 1.00 m3 of an ideal gas at this temperature and pressure? b. Avogadros number of air molecules has a mass of 2.85 102 kg. What is the mass of 1.00 m3 of air? c. Does the value calculated in part (b) agree with the stated density of air at this temperature?arrow_forwardFor a temperature increase of 10 at constant volume, what is the heat absorbed by (a) 3.0 mol of a dilute monatomic gas; (b) 0.50 mol of a dilute diatomic gas; and (c) 15 mol of a dilute polyatomic gas?arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forward
- An ideal gas initially at 300 K undergoes an isobaric expansion at 2.50 kPa. If the volume increases from 1.00 m3 to 3.00 m3 and 12.5 kJ is transferred to the gas by heat, what are (a) the change in its internal energy and (b) its final temperature?arrow_forwardA 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forwardOne process for decaffeinating coffee uses carbon dioxide ( M=44.0 g/mol) at a molar density of about 14,0 mol/m3 and a temperature of about 60 . (a) Is CO2 a solid, liquid, gas, or supercritical fluid under those conditions? (b) The van der Waals constants for carbon dioxide are a=0.3658 Pa m6/mol2 and b=4.286105 m3/mol. Using the van der Waals equation, estimate pressure of CO2 at that temperature and density. `arrow_forward
- Cylinder A contains oxygen (O2) gas, and cylinder B contains nitrogen (N2) gas. If the molecules in the two cylinders have the same rms speeds, which of the following statements is false? (a) The two gases haw different temperatures. (b) The temperature of cylinder B is less than the temperature of cylinder A. (c) The temperature of cylinder B is greater than the temperature of cylinder A. (d) The average kinetic energy of the nitrogen molecules is less than the average kinetic energy of the oxygen molecules.arrow_forwardA 1.00-mol sample of hydrogen gas is heated at constant pressure from 300 K to 420 K. Calculate (a) the energy transferred to the gas by heat, (b) the increase in its internal energy, and (c) the work done on the gas.arrow_forwardOne mole of an ideal gas does 3 000 J of work on its surroundings as it expands isothermally to a final pressure of 1.00 atm and volume of 25.0 L. Determine (a) the initial volume and (b) the temperature of the gas.arrow_forward
- A sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P21.65). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state, (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally, (f) Find Q, W, and Eint for each of the processes, (g) For the whole cycle A B C A, find Q, W, and Eint.arrow_forwardIf a gas is compressed isothermally, which of the following statements is true? (a) Energy is transferred into the gas by heat. (b) No work is done on the gas. (c) The temperature of the gas increases. (d) The internal energy of the gas remains constant. (e) None of those statements is true.arrow_forward1.50 moles of a monatomic ideal gas goes isothermally from state 1 to state 2. P1 = 2.8×105 Pa, V1 = 88 m3, and P2 = 6.6×105 Pa. What is the volume in state 2, in m3? Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement.arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning



