
Concept explainers
Acid-catalyzed hydrolysis of a nitrile to give a carboxylic acid occurs by initial protonation of the nitrogen atom, followed by nucleophilic addition of water. Review the mechanism of base-catalyzed nitrile hydrolysis in Section 20-7 and then predict the products for each reaction below and write all of the steps involved in the acid-catalyzed reaction, using curved arrows to represent electron flow in each step.
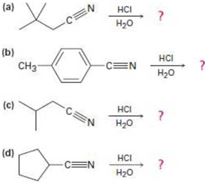
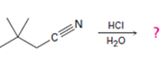
a)
Interpretation:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step, is to be given.
Concept introduction:
The acid protonates the nitrogen of the nitrile group initially. The nucleophilic attack by water and subsequent proton transfer will yield an intermediate. Another nucleophilic attack by water on the intermediate and yet another proton transfer produces another intermediate. The intermediate loses ammonia to produce the protonated acid which deprotonates to yield the acid.
To give:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step.
Answer to Problem 25MP
The products of the reaction are ammonia and 2,2-dimethylbutanoic acid.
The mechanism of the reaction is given below.
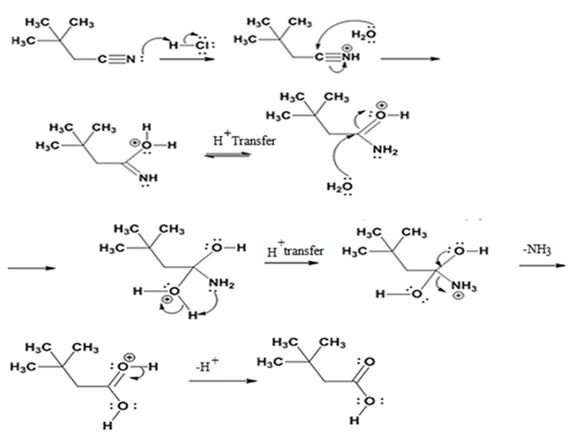
Explanation of Solution
In the first step, 2,2-dimethylbutane nitrile is protonated by HCl. In the next step, the nucleophilic attack of water on the protonated nitrile occurs and the accompanying proton transfer yields a protonated aminoketone. Another nucleophilic attack on the carbonyl carbon of the protonated aminoketone in the next step and the subsequent proton transfer yields a protonated diol intermediate which eliminates ammonia and a proton in the subsequent steps to yield 2,2-dimethylbutanoic acid.
The products of the reaction are ammonia and 2,2-dimethylbutanoic acid.
The mechanism of the reaction is given below.

b)
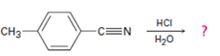
Interpretation:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step, is to be given.
Concept introduction:
The acid protonates the nitrogen of the nitrile group initially. The nucleophilic attack by water and subsequent proton transfer will yield an intermediate. Another nucleophilic attack by water on the intermediate and yet another proton transfer produces another intermediate. The intermediate loses ammonia to produce the protonated acid which deprotonates to yield the acid.
To give:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step.
Answer to Problem 25MP
The products of the reaction are ammonia and p-methylbenzoic acid.
The mechanism of the reaction is given below.
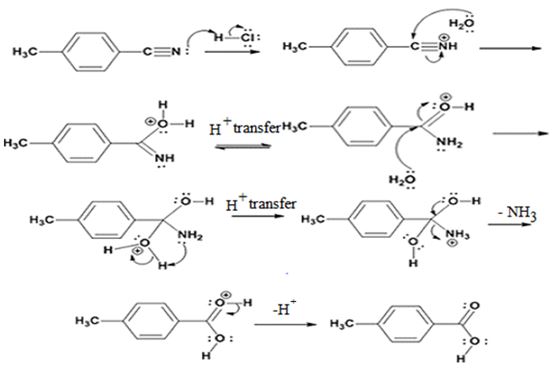
Explanation of Solution
In the first step, p-methylbenzonitrile is protonated by HCl. In the next step, the nucleophilic attack of water on the protonated nitrile occurs and the accompanying proton transfer yields a protonated aminoketone. Another nucleophilic attack on the carbonyl carbon of the protonated aminoketone in the next step and the subsequent proton transfer yields a protonated diol intermediate which eliminates ammonia and a proton in the subsequent steps to yield p-methylbenzoic acid.
The products of the reaction are ammonia and p-methylbenzoic acid.
The mechanism of the reaction is given below.

c)
Interpretation:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step, is to be given.
Concept introduction:
The acid protonates the nitrogen of the nitrile group initially. The nucleophilic attack by water and subsequent proton transfer will yield an intermediate. Another nucleophilic attack by water on the intermediate and yet another proton transfer produces another intermediate. The intermediate loses ammonia to produce the protonated acid which deprotonates to yield the acid.
To give:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step.
Answer to Problem 25MP
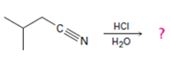
The products of the reaction are ammonia and 2-methylbutanoic acid.
The mechanism of the reaction is given below.
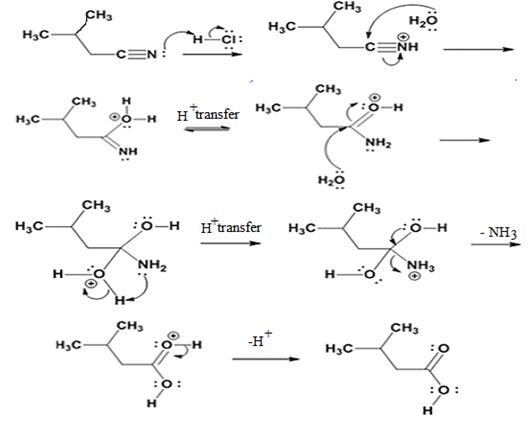
Explanation of Solution
In the first step, 2-methylbutane nitrile is protonated by HCl. In the next step, the nucleophilic attack of water on the protonated nitrile occurs and the accompanying proton transfer yields a protonated aminoketone. Another nucleophilic attack on the carbonyl carbon of the protonated aminoketone in the next step and the subsequent proton transfer yields a protonated diol intermediate which eliminates ammonia and a proton in the subsequent steps to yield 2-methylbutanoic acid.
The products of the reaction are ammonia and 2-methylbutanoic acid.
The mechanism of the reaction is given below.

d)
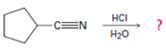
Interpretation:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step, is to be given.
Concept introduction:
The acid protonates the nitrogen of the nitrile group initially. The nucleophilic attack by water and subsequent proton transfer will yield an intermediate. Another nucleophilic attack by water on the intermediate and yet another proton transfer produces another intermediate. The intermediate loses ammonia to produce the protonated acid which deprotonates to yield the acid.
To give:
The products of the reaction, the acid-catalyzed nitrile hydrolysis, along with all the steps involved using curved arrows to represent electron flow in each step.
Answer to Problem 25MP
The products of the reaction are ammonia and cyclopentanecarboxylic acid.
The mechanism of the reaction is given below.
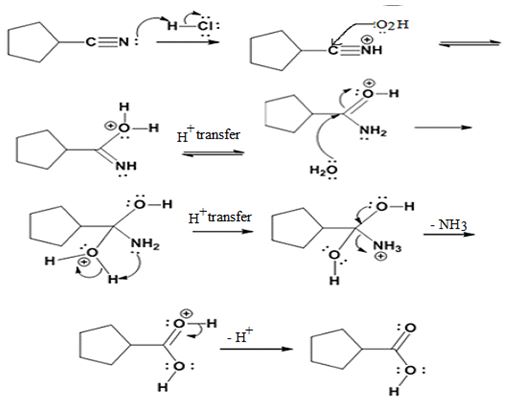
Explanation of Solution
In the first step, cyclopentanenitrile is protonated by HCl. In the next step, the nucleophilic attack of water on the protonated nitrile occurs and the accompanying proton transfer yields a protonated aminoketone. Another nucleophilic attack on the carbonyl carbon of the protonated ketone in the next step and the subsequent proton transfer yields a protonated aminodiol intermediate which eliminates ammonia and a proton in the subsequent steps to yield cyclopentanecarboxylic acid.
The products of the reaction are ammonia and cyclopentanecarboxylic acid.
The mechanism of the reaction is given below.

Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry
- Enamines normally react with methyl iodide to give two products: one arising from alkylation at nitrogen and the second arising from alkylation at carbon. For example, Heating the mixture of C-alkylation and N-alkylation products gives only the product from C-alkylation. Propose a mechanism for this isomerization.arrow_forwardOne frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forwardH NH₂ ཡིནྣཾ ༥ ཨ ཨནྡྷ༥ ༠ ཨི་ཝཱ, ཙ ཨ་ར༩ H NH3+ the acidity of the amine ion drives the reaction to shift toward the conjugate base of the carboxylic acid product. the resulting carboxylic acid ion is a weaker base than an acetate ion. O aldehydes are more reactive toward nucleophiles than ketones. Onucleophilic attack occurs preferentially at the less hindered carbon of the formyl group.arrow_forward
- Complete the following proposed acid–base reactions, and predict whether the reactants or products are favored.arrow_forwardH3C OH OH H+ CH3 Esters can be synthesized by an acid-catalyzed nucleophilic acyl substitution between an alcohol and a carboxylic acid; this process is called the Fischer esterification reaction. Because the alcohol oxygen is a poor nucleophile, the carbonyl carbon is made a better electrophile by protonation of the carbonyl oxygen. The steps of the synthesis are all reversible. The reaction is generally driven to completion by using an excess of the liquid alcohol as a solvent, or by distilling off the product as it forms. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions CIX 10-4 H₂O CH3 H₂O CH3arrow_forwardPh3P-CHCH3 H₂C H3C Aldehydes and ketones are converted into alkenes by means of a direct nucleophilic addition called the Wittig reaction. In the reaction, a triphenylphosphorine ylide, also called a phosphorane, adds to an aldehyde/ketone to give a four-membered cyclic intermediate called an oxaphosphetane. The oxaphosphetane is not isolated but instead spontaneously decomposes to release triphenylphosphine oxide and an alkene. CH3 00 + The ylide is formed by reaction of triphenylphosphine, a good nucleophile, with a primary alkyl halide in an SN2 reaction, followed by deprotonation of the carbon with a strong base, such as butyllithium. The carbonyl carbon and the carbon originally bonded to the halogen become the two carbons with the double bond in the product alkene X m CH3 The real value of the Wittig reaction lies in its ability to yield an alkene of predictable structure, as the C=C bond is precisely where the C=O bond was in the reactant and no isomers (other than E/Z isomers)…arrow_forward
- what is the best way to come up with how to synthesize carboxylic acid , oct-4-yne to butanoic acid, or trans cyclodecene to decanedoic acid . I keep getting confused on how to go from the reactants to product, even with the amines. i can't figure out what to use to get to the product. Please explain Grignard rx as well.arrow_forwardShow how acid derivatives hydrolyze to carboxylic acids under either acidic or basicconditions. Explain why some acid derivatives (amides, for example) require muchstronger conditions for hydrolysis than other derivatives.arrow_forwardCH3 Ph3P-CHCH3 H3C H3C Aldehydes and ketones are converted into alkenes by means of a direct nucleophilic addition called the Wittig reaction. In the reaction, a triphenylphosphorine ylide, also called a phosphorane, adds to an aldehyde/ketone to give a four-membered cyclic intermediate called an oxaphosphetane. The oxaphosphetane is not isolated but instead spontaneously decomposes to release triphenylphosphine oxide and an alkene. Ph3P-CHCH3 H3C The ylide is formed by reaction of triphenylphosphine, a good nucleophile, with a primary alkyl halide in an S 2 reaction, followed by deprotonation of the carbon with a strong base, such as butyllithium. The carbonyl carbon and the carbon originally bonded to the halogen become the two carbons with the double bond in the product alkene :0: CH3 Com The real value of the Wittig reaction lies in its ability to yield an alkene of predictable structure, as the C-C bond is precisely where the C=O bond was in the reactant and no isomers (other than…arrow_forward
- Succinic acid can be synthesized by the following series of reactions from acetylene. Show the reagents and experimental conditions necessary to carry out this synthesis. НО OH НО. HO H- H. HO, ОН Acetylene 2-Butyne-1,4-diol 1,4-Butanediol Butanedioic acid (Succinic acid)arrow_forwardHydration of aldehydes and ketones can be catalyzed by acid or base. Bases catalyze hydration by: protonating the carbonyl oxygen making the carbonyl group more electrophilic employing hydroxide ion, which is a better nucleophile than water making the carbonyl group less electrophilic shifting the equilibrium position of the reaction to favor productsarrow_forwardb) The Wolf-Kishner reduction is a reaction used in Organic Chemistry to convert carbonyl functionalities into methylene group. The reaction was used to convert an aldehyde or ketone to an alkane using hydrazine, base and thermal conditions. The mechanism begins with the attack of hydrazine of the aldehyde or ketone. Stage 1: The reaction of aldehyde/ketone with hydrazine to produce hydrazine Stage 2: Reaction with the base and heat to convert hydrozone to alkane Write the mechanism of the reaction.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


