
(a)
To draw a PV diagram for the gas.
(a)
Answer to Problem 42PQ
The PV diagram for the gas is given below.
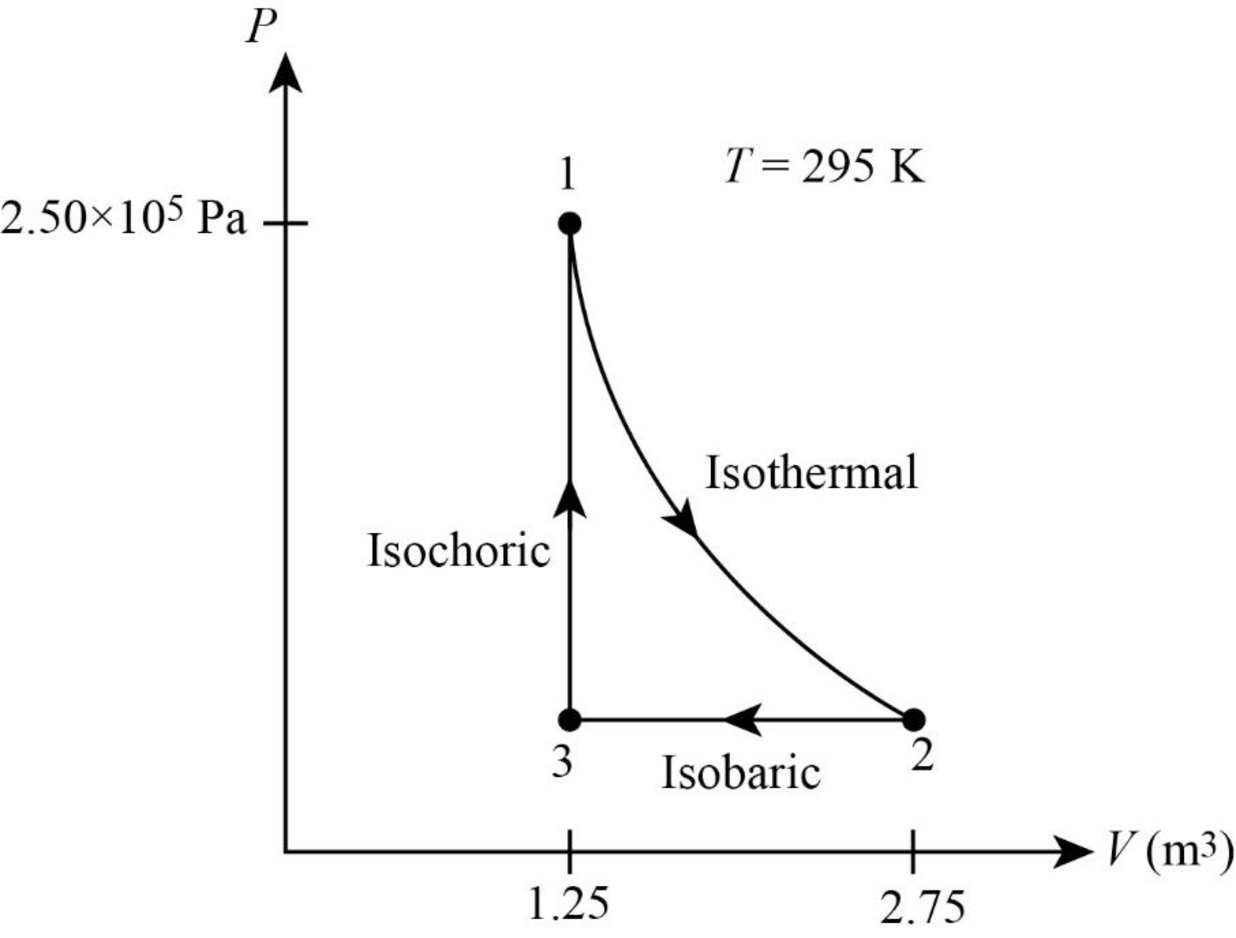
Explanation of Solution
It is given that pressure of the gas is
The following figure gives the PV diagram for the gas.
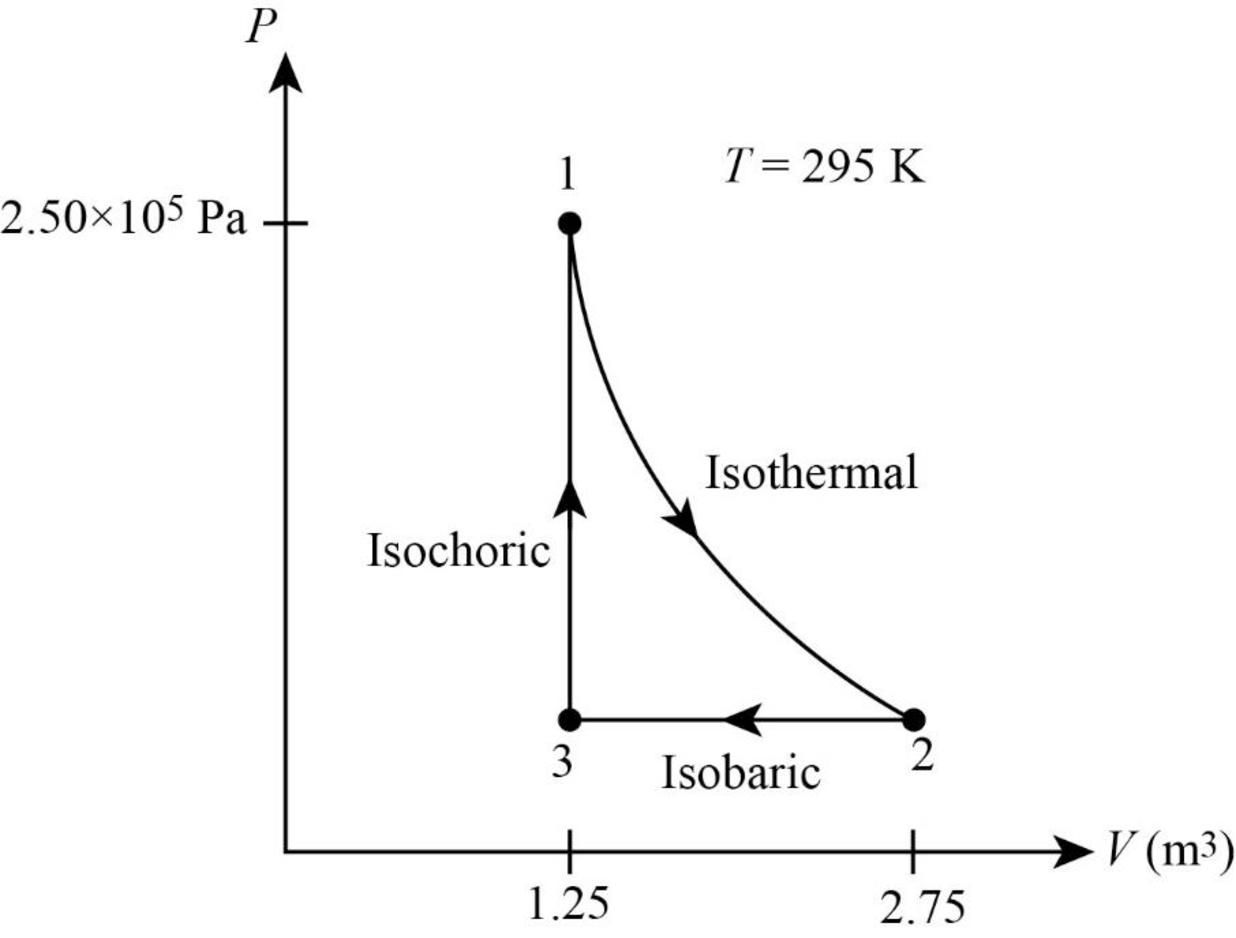
In above figure, point 1 represents the initial state. The gas follows isothermal expansion from
Conclusion:
Thus, the PV diagram for the gas is given below.
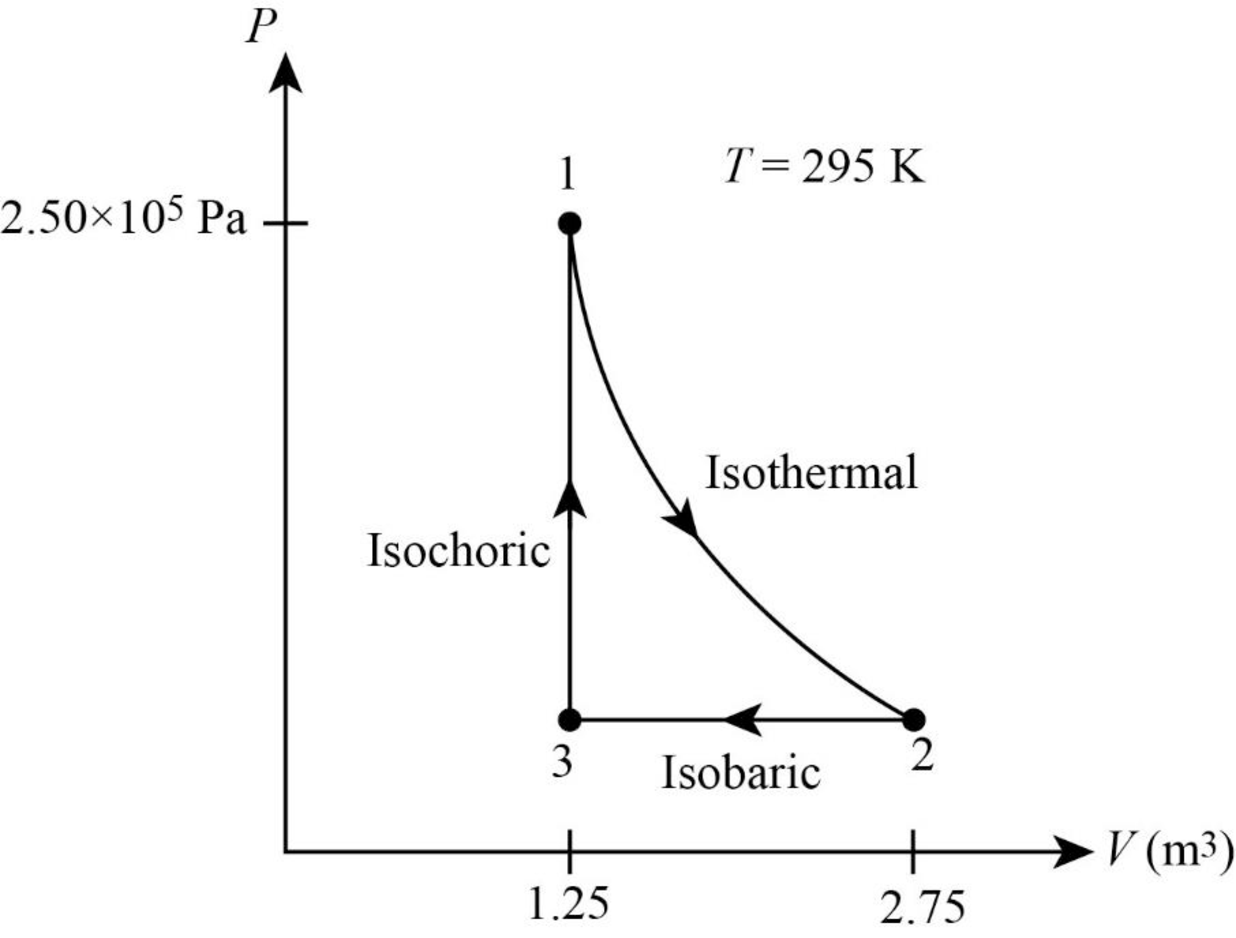
(b)
The change in thermal energy.
(b)
Answer to Problem 42PQ
The change in thermal energy of the gas is zero.
Explanation of Solution
From figure its clear that the gas undergoes a
In this problem it is given that the system return’s to its original state. Thus following a cyclic process, total change in thermal energy of the gas is zero.
Conclusion:
Therefore, the change in thermal energy of the gas is zero.
(c)
The work done by environment on the gas.
(c)
Answer to Problem 42PQ
The work done by environment on the gas is
Explanation of Solution
The net work done by the gas is the area inside the curve. In isothermal process volume of the system is raised from
Write the expression for the work done during isothermal expansion.
Here,
Write ideal gas equation.
Here,
Use above equation for the gas at initial state (state 1 in figure).
Here,
Substitute
For isothermal process,
Apply above equation for the isothermal process of the gas.
Here,
Rearrange above equation to get
In figure1, the straight line represents isobaric process where pressure is constant. During this process the system is compressed to original volume. Therefore, work is done on the system.
Write the expression for the work done in isobaric compression.
Here,
The negative sign indicates that work is done on the gas.
During isobaric process, the gas is at pressure
Use above equation to write work done by gas during isobaric compression shown in figure1.
Here,
In an isochoric process total work done is zero.
Write the expression for the total work done by the gas.
Here,
Conclusion:
Substitute
Substitute
Substitute
Substitute
Therefore, the work done by environment on the gas is
(d)
The heat that flows into the gas.
(d)
Answer to Problem 42PQ
The heat that flows into the gas is
Explanation of Solution
Write the first law of
Here,
Since the process is cyclic,
Substitute
Conclusion:
Substitute
Therefore, the heat that flows into the gas is
Want to see more full solutions like this?
Chapter 21 Solutions
Physics for Scientists and Engineers: Foundations and Connections
- In a cylinder of an automobile engine, immediately after combustion the gas is confined to a volume of 50.0 cm3 and has an initial pressure of 3.00 106 Pa. The piston moves outward to a final volume of 300 cm3, and the gas expands without energy transfer by heat, (a) What is the final pressure of the gas? (b) How much work is done by the gas in expanding?arrow_forwardA 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forwardAn ideal gas is compressed to half its initial volume by means of several possible processes. Which of the following processes results in the most work done on the gas? (a) isothermal (b) adiabatic (c) isobaric (d) The work done is independent of the process.arrow_forward
- (a) An ideal gas expands adiabatically from a volume of 2.0103 m3 to 2.5103 m3. If the initial pressure and temperature 5.0105 Pa and 300 K, respectively, what are the final pressure and temperature of the gas? Use =5/3 for the gas. (b) In an isothermal process, an ideal gas expands from a of 2.0103 m3 to 2.5103 m3. If the initial pressure and temperature were 5.0105 Pa and 300 K, respectively, what are the final pressure and temperature of the gas?arrow_forward(a) How much food energy will a man metabolize in the process of doing 35.0 kJ of work with an efficiency of 5.00%? (b) How much heal transfer occurs to the environment to keep his temperature constant? Explicitly show how you follow the steps in the Problem—Solving Strategy for thermodynamics found in Problem-Solving Strategies for Thermodynamics.arrow_forward(a) Calculate the rate of heat transfer by radiation from a car radiator at 110C into a 50.0C environment, if the radiator has an emissivity of 0.750 and a 1.20m2 surface area. (b) Is this a significant fraction of the heat transfer by an automobile engine? To answer this, assume a horsepower of 200 hp (1.5 kW) and the efficiency of automobile engines as 25%.arrow_forward
- In a cylinder, a sample of an ideal gas with number of moles n undergoes an adiabatic process. (a) Starting with the expression W=PdV and using the condition PV = constant, show that the work done on the gas is W=(11)(PfVfPiVi) (b) Starting with the first law of thermodynamics, show that the work done on the gas is equal to nCV(Tf Ti). (c) Are these two results consistent with each other? Explain.arrow_forwardA gas expands from I to Fin Figure P20.58 (page 622). The energy added to the gas by heat is 418 J when the gas goes from I to F along the diagonal path, (a) What is the change in internal energy of the gas? (b) How much energy must be added to the gas by heat along the indirect path IAF?arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





