
Concept explainers
Interpretation:
The Lewis diagram of the general structure of an amino acid is to be drawn.
Concept introduction:
The Lewis diagram shows the bonding between different atoms of a molecule and the lone pairs of electrons in the molecule. It shows how the valence electrons of an atom are arranged in the molecule. In Lewis diagram, two electrons between the atoms are indicated as a line to show bonding between atoms.
Answer to Problem 1E
The Lewis diagram of the general structure of an amino acid is shown below.
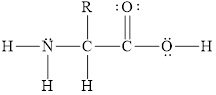
Explanation of Solution
The Lewis structure of a compound shows how the valence electrons of an atom are arranged in the molecule. The molecular structures are given by its valence electrons and lone pairs on central atom.
The total number of valence electrons in an amino acid is the sum of valence electrons of two carbon atoms, valence electrons of four hydrogen atoms, valence electrons of one nitrogen atom and valence electron of two oxygen atoms. There are four valence electrons on carbon atom, one valence electron on each hydrogen atom, five valence electrons on nitrogen atom and six valence electrons on each oxygen atom. Therefore, the total number of valence electron in an amino acid is

Figure 1
The Lewis diagram of the general structure of an amino acid is shown in Figure 1.
Want to see more full solutions like this?
Chapter 22 Solutions
Introductory Chemistry: An Active Learning Approach
- What functional groups are found in all amino acids? How many different amino acids are found in naturally occurring proteins?arrow_forwardWhich of the following are true concerning the chemical bond that forms between the carboxyl (RCOOH) group of one amino acid and the amino (RCNH2) group of another? a.The bond is called a peptide bond. b.It is formed by inserting a water molecule between them. c.It is formed by a dehydration reaction. d.A polypeptide has more of these bonds than a protein.arrow_forwardWhat is the basis for deciding if a substance is a lipid?arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





