
(a)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
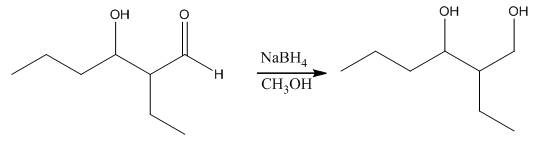
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown as,
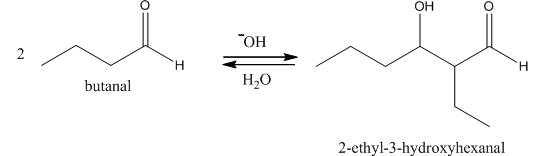
Figure 1
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert

Figure 2
Thus, the reagent that is needed to convert
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(b)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
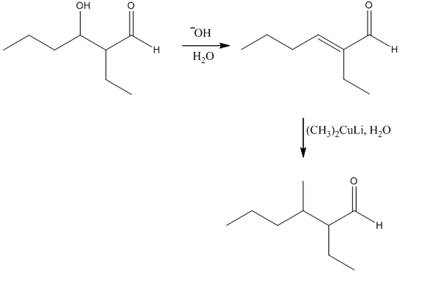
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert
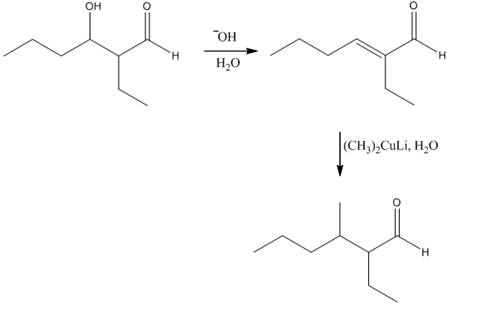
Figure 3
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(c)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
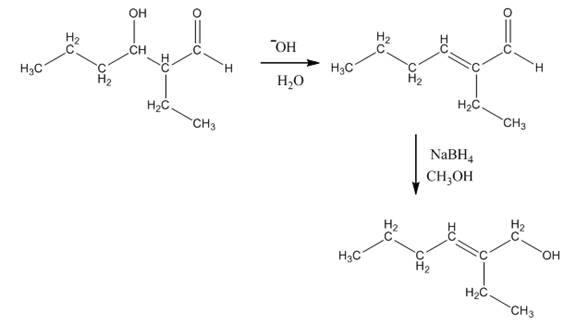
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert
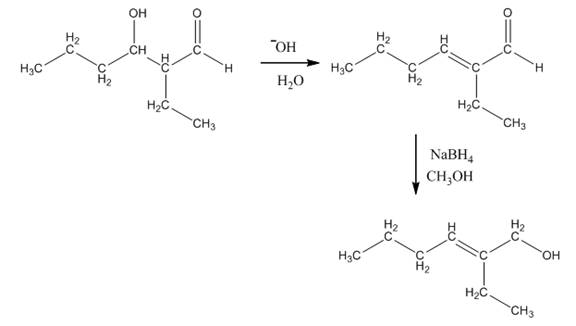
Figure 4
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(d)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
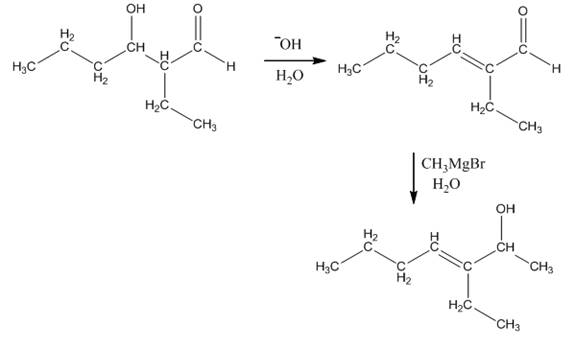
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert

Figure 5
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- What aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the attached compounds?arrow_forward1. how to synthesize a and barrow_forward1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forward
- Identify the following ethers or epoxides as symmetrical or asymmetrical. а. b. С. a.arrow_forwardDraw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form asubstituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that canbe used as a synthetic detergent to clean away dirt.arrow_forwardProblem 15 of 44 68 Draw the major product of this reaction. Ignore inorganic byproducts. TSOH, CH3CH2OH (2 equiv) Submit Assume that the water side product is continuously removed to drive the reaction toward products. Select to Drawarrow_forward
- Identify the missing reagents a-f in the following scheme:arrow_forwardDraw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that can be used as a synthetic detergent to clean away dirt.arrow_forwardWhich is the major product?a. Ab. Neither product would likely formc. Barrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


