
Concept explainers
Describe what is meant by each of the following reaction types, and illustrate with an example:
(a) nucleophilic substitution reaction: (b) electrophilic substitution reaction; (c) addition reaction;
(d) elimination reaction, (e) rearrangement reaction.
(a)
Interpretation:
The nucleophilic substitution reaction should be defined with example.
Concept introduction:
Nucleophilic substitution reaction describes the attack of the electron-rich group that is nucleophile on electron deficient groups that is electrophile.
Answer to Problem 1E
Nucleophilic substitution reaction is the type of reaction in which the nucleophile (electron rich species) attacks the electron-deficient carbon atom which is electrophilic.
Explanation of Solution
Nucleophilic substitution reaction is defined as an organic reaction which includes the attack of a nucleophile on electrophilic center along with the removal of the leaving group.
The example of the nucleophilic substitution reaction is,
In this reaction, chlorine of chloroethane is replaced by a hydroxyl group
(b)
Interpretation:
The electrophilic substitution reaction should be defined with example.
Concept introduction:
The electrophilic substitution reaction describes the displacement of functional group or hydrogen atom by an electron deficient group or electrophile.
Answer to Problem 1E
Electrophilic substitution reaction is defined as the organic reaction in which the electrophile replaces a functional group of a compound or hydrogen atom.
Explanation of Solution
Electrophilic substitution reaction is defined as the organic reaction which includes the replacement of functional group or
Example of electrophilic substitution reaction is,
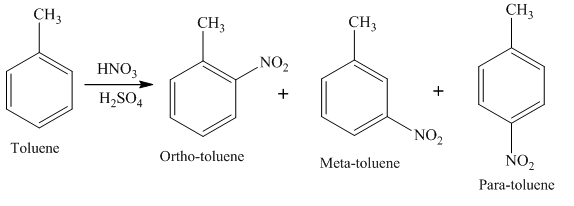
In this reaction, Toluene undergoes electrophilic substitution to form para nitrotoluene, meta nitrotoluene, and ortho nitrotoluene.
(c)
Interpretation:
The addition reaction should be defined with example.
Concept introduction:
The addition reaction describes the combination of two or more smaller molecules to form a larger molecule.
Answer to Problem 1E
Addition reaction is defined as the reaction in which two or more molecules combine to form a single and large molecule.
Explanation of Solution
The reaction of the addition of the two or more reactants that is A and B to produce a single product C is termed as addition reaction.
The example of addition reaction is,
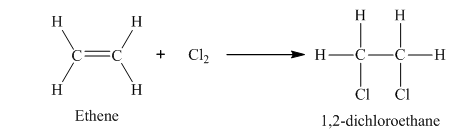
In this reaction, chlorine molecule combines with ethene to form 1, 2-dichloroethane.
(d)
Interpretation:
The elimination reaction should be defined with example.
Concept introduction:
The elimination reaction describes the removal of two substituents from the reactant molecule to form the product.
Answer to Problem 1E
Elimination reaction is the reaction by which the reactant molecule or compound breaks into two or more products.
Explanation of Solution
Elimination reaction is the type of reaction in which two substituents are removed from the reactant molecule to form the product. Generally, unsaturated compounds are formed in an elimination reaction.
The example of the elimination reaction is,
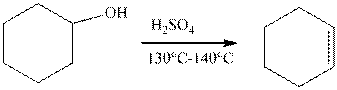
The reaction of cyclohexanol in the presence of
(e)
Interpretation:
The rearrangement reaction should be defined with example.
Concept introduction:
The rearrangement reaction describes the rearrangement of bonds in a molecule to form the product.
Answer to Problem 1E
It is the process of movement of bonds within a molecule to give rise to structural isomers.
Explanation of Solution
It is defined as a reaction in which an atom or a bond migrates from one atom in reactant molecule to adjacent atom to give rise to the product.
Example of rearrangement reaction is,
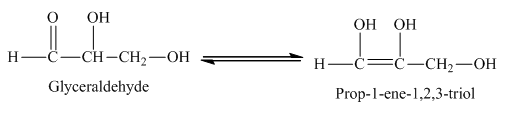
The glyceraldehyde undergoes rearrangement to form enediol.
Want to see more full solutions like this?
Chapter 27 Solutions
GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
Additional Science Textbook Solutions
Organic Chemistry (9th Edition)
Chemistry In Context
Chemistry For Changing Times (14th Edition)
EBK INTRODUCTION TO CHEMISTRY
Elementary Principles of Chemical Processes, Binder Ready Version
- How can alkenes and alkynes be classified: as nucleophiles, as electrophiles or depending on the reaction conditions could be both? Explain extensively.arrow_forward1. Draw the organic product formed when the following compounds undergo a substitution reaction ... : (a) acetic acid and methylamine; (b) butanoic acid and 2-propanol; (c) formic acid and 2-methyl- 1-propanol 2. Draw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and 1- hexanol; (b) propanoic acid and dimethylamine; (c) ethanoic acid and diethylamine.arrow_forward(a) Draw a specific organic reaction for the hydration addition reaction of alkenes. (b) Also write the reagents involved in the reaction (c) and name and identify its major and minor product by writing its IUPAC name.arrow_forward
- (a) Draw a specific organic reaction for the halogenation addition reaction of alkenes. (b) Also write the reagents involved in the reaction (c) and name and identify its major and minor product by writing its IUPAC name.arrow_forward4. Outline the following synthesis (a). Butane to 2-butanol (b). 1,2-dibromopropane from 2-propanol (c). 1-butanol to 2-butanol (d). Propane to Propenearrow_forwardWhich of the following compounds, is the most acidic: (a) 2-methylhex-3-yne, (b) 2,5-dimethylhex-3-yne or (c) 2,2,5-trimethylhex-3-yne?arrow_forward
- Draw the structures for each of the following molecules. (a) 2-fluorotoluene; (b) 4-ethoxytoluene; (c) 2-ethoxyanisole;(d) 1,3-diphenylheptane; (e) 4,4-diphenyl-1-octene; (f) benzylbenzenearrow_forwardDraw the structure of each of the following molecules. (a) pentanoic anhydride; (b) hexanoic propanoic anhydride; (c) ethanoic 3-methylpentanoic anhydridearrow_forward5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forward
- 6. Dacron is the brand name for the polymer that is made from ethane-1,2-diol and benzene-1,4-dicarboxylic acid. (a) When the two monomers combine, what type of reaction do they undergo and what molecule is eliminated? [1] (b) What is the name for the linkages that join the monomers together? [1] (c) Draw the monomers and the polymer of the reaction to create Dacron. [3]arrow_forwardGive the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forward(a) Write a chemical test to distinguish between: (i) Chlorobenzene and Benzyl chloride. (ii) Chloroform and Carbon tetrachloride. (b) Why is methyl chloride hydrolysed more easily than chlorobenzene?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





