
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 2.7, Problem 2.26E
Interpretation Introduction
Interpretation: Zigzag hashed-wedged line structures for
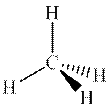
Concept introduction: The wedge bond depicts a bond that comes out of plane of paper, and the dash bond denotes bond that goes behind the plane of paper. This representation is used for correct three-dimensional representation of organic substrates as otherwise it might be difficult to visualize the orientation of different bonds in space. For example in methane 3D representation on plane two bonds in plane are shown by usual bonds, while one behind the plane and above the plane are denoted by dash and wedge type bonds as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following is a correct line structure for 3-ethyl-3-methylpentane?
How many asymmetric carbons are present in Ethyl-2,2,4-tnmethypentane? Indicate them.
Indicate for the following compound: (1) the number of carbons in the
longest carbon chain, (2) the positions of the double bonds [separate # with
comma], (3) the IUPAC name of the compound.
Answer for blank # 1: 8
Answer for blank # 2: 2,5
Answer for blank # 3: 2,5-dimethyl
Chapter 2 Solutions
Organic Chemistry: Structure and Function
Ch. 2.1 - Prob. 2.1ECh. 2.1 - Prob. 2.2ECh. 2.1 - Prob. 2.3ECh. 2.1 - Prob. 2.5TIYCh. 2.1 - Prob. 2.6ECh. 2.1 - Prob. 2.7ECh. 2.2 - Prob. 2.8ECh. 2.3 - Prob. 2.9ECh. 2.3 - Prob. 2.10ECh. 2.3 - Prob. 2.11E
Ch. 2.3 - Prob. 2.12ECh. 2.3 - Prob. 2.13ECh. 2.3 - Prob. 2.14ECh. 2.3 - Prob. 2.15ECh. 2.3 - Prob. 2.17TIYCh. 2.3 - Prob. 2.19TIYCh. 2.5 - Prob. 2.20ECh. 2.6 - Prob. 2.21ECh. 2.6 - Prob. 2.22ECh. 2.6 - Prob. 2.23ECh. 2.6 - Prob. 2.25TIYCh. 2.7 - Prob. 2.26ECh. 2.9 - Prob. 2.28TIYCh. 2 - Prob. 31PCh. 2 - Prob. 32PCh. 2 - Prob. 33PCh. 2 - Prob. 34PCh. 2 - Prob. 35PCh. 2 - Prob. 36PCh. 2 - Prob. 37PCh. 2 - Prob. 38PCh. 2 - Prob. 39PCh. 2 - Prob. 40PCh. 2 - Prob. 41PCh. 2 - Prob. 42PCh. 2 - Prob. 43PCh. 2 - Prob. 44PCh. 2 - Prob. 45PCh. 2 - Prob. 46PCh. 2 - Prob. 47PCh. 2 - Prob. 48PCh. 2 - Prob. 49PCh. 2 - Prob. 50PCh. 2 - Prob. 51PCh. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - Prob. 56PCh. 2 - Prob. 57PCh. 2 - Prob. 58PCh. 2 - Prob. 59PCh. 2 - Prob. 60PCh. 2 - Prob. 61PCh. 2 - Prob. 62PCh. 2 - Prob. 63PCh. 2 - Prob. 64PCh. 2 - Prob. 65PCh. 2 - Prob. 66P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following is not another representation for 2-methylbutane?arrow_forwardMonochlorination reaction of the following alkane gave mixtures of isomers product. Which of the following is NOT the correct isomer? Cl, hv, 25°C CI CIarrow_forward(iii) In the presence of a suitable catalyst, 2-methylpropene forms a mixture of dimers. Two of these dimers react with hydrogen to form * 2,2,4-trimethylpentane. Draw the skeletal formula for 2,2,4-trimethylpentane. Use this to draw the skeletal structure of one of the dimers formed from 2-methylpropene. 2,2,4-trimethylpentane Dimerarrow_forward
- Draw the structure of trans-1-cis-Butyl-4-methylcyclohexane. Show lone pairs and charges.arrow_forwardName the alkyl group as if the dashed line were where it was connect to the rest of the molecule, state whether it is primary, secondary, or tertiary.arrow_forwardskeletal bond line drawing of 2-methylheptanearrow_forward
- Consider 1,2-dimethylcyclohexane.Q: Draw structures for the cis and trans isomers using a hexagon for the sixmembered ring.arrow_forwardDraw the bond-line formula/structure of cis-4-methyl-2-hexene and trans-4-methyl-2-hexene.arrow_forwardHow could you show experimentally that the molecular formula of propene is C3H6, not CH2?arrow_forward
- Draw an alkane containing one tertiary carbon atom, one secondary carbon atom, and three primary carbon atoms. Show all hydrogen atoms.arrow_forwardThe following name given is incorrect or incomplete, but it represents a real structure. Draw structure and name it correctly.(a) 2-dimethylbutanearrow_forwardName the following molecule. • Draw the bond-line representation for 1-tert-butyl-2-ethyl-4-methyl-3- cyclopentylcyclohexane.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License