
(a)
Interpretation:
The structure of the product expected when valine would react with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with
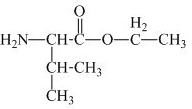
The structure of the product expected when proline would react with
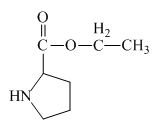
Explanation of Solution
The valine has a carboxylic group in it. This carboxylic group can undergo esterification with ethanol in the presence of sulfuric acid. The corresponding
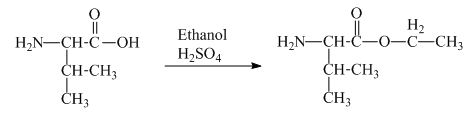
Figure 1
The proline has a carboxylic group in it. This carboxylic group can undergo esterification with ethanol in the presence of sulfuric acid. The corresponding chemical reaction is shown below.
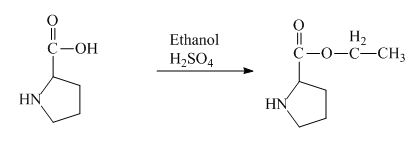
Figure 2
The structure of the product expected when valine would react with
(b)
Interpretation:
The structure of the product expected when valine would react with benzoyl chloride and
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with benzoyl chloride and ![]() is shown below.
is shown below.
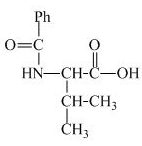
The structure of the product expected when proline would react with benzoyl chloride and
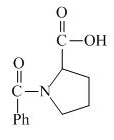
Explanation of Solution
The valine has a basic
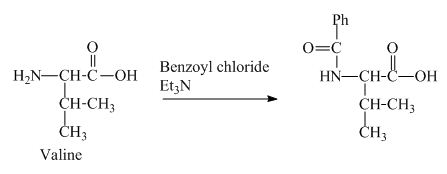
Figure 3
The proline has a basic amine group in it. This group can react with benzoyl chloride to form amide bond. The corresponding chemical reaction is shown below.
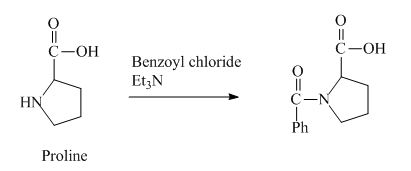
Figure 4
The structure of the product expected when valine would react with benzoyl chloride and
The structure of the product expected when proline would react with benzoyl chloride and
(c)
Interpretation:
The structure of the product expected when valine would react with an aqueous
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with an aqueous
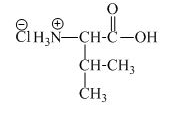
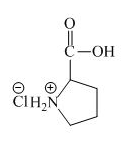
The structure of the product expected when proline would react with an aqueous
Explanation of Solution
The valine has a basic amine group in it. This amine group can react with acid to form an ammonium base. The corresponding chemical reaction is shown below.
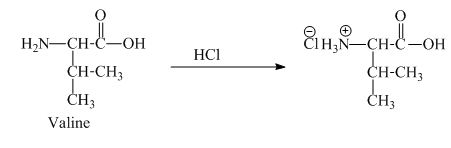
Figure 5
The proline has a basic amine group in it. This amine group can react with acid to form an ammonium base. The corresponding chemical reaction is shown below.
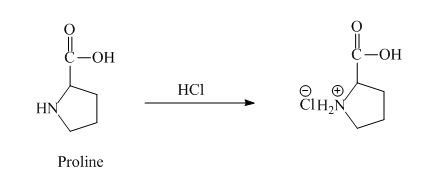
Figure 6
The structure of the product expected when valine would react with an aqueous
The structure of the product expected when proline would react with an aqueous
(d)
Interpretation:
The structure of the product expected when valine would react with an aqueous
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with an aqueous
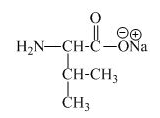
The structure of the product expected when proline would react with an aqueous
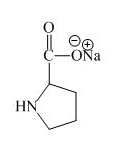
Explanation of Solution
The valine has an acidic carboxylic group in it. This group can react with a base such as sodium hydroxide to form a sodium salt of carboxylic acid. The corresponding chemical reaction is shown below.
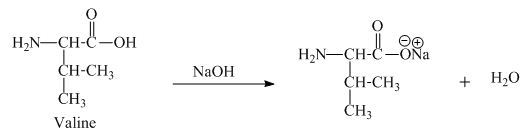
Figure 7
The proline has an acidic carboxylic group in it. This group can react with a base such as sodium hydroxide to form a sodium salt of carboxylic acid. The corresponding chemical reaction is shown below.
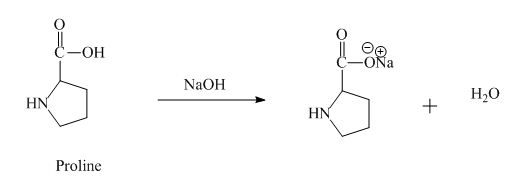
Figure 8
The structure of the product expected when valine would react with an aqueous
The structure of the product expected when proline would react with an aqueous
(e)
Interpretation:
The structure of the product expected when valine would react with aqueous benzaldehyde and
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with aqueous benzaldehyde and
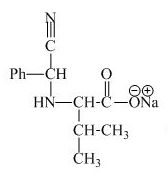
The structure of the product expected when proline would react with aqueous benzaldehyde and
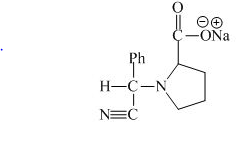
Explanation of Solution
The valine has a basic group in it. This group can be attacked by the benzaldehyde in the presence of sodium cyanide to form the final product. The corresponding chemical reaction is shown below.
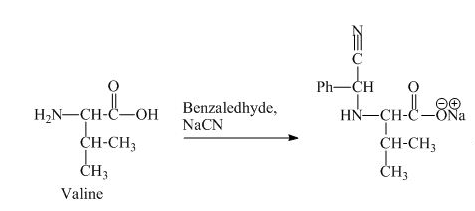
Figure 9
The proline has a basic group in it. This group can be attacked by the benzaldehyde in the presence of sodium cyanide to form the final product. The corresponding chemical reaction is shown below.
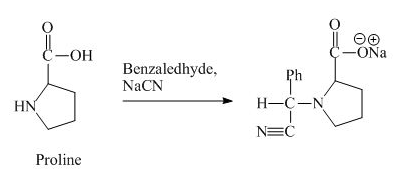
Figure 10
The structure of the product expected when valine would react with aqueous benzaldehyde and
The structure of the product expected when proline would react with aqueous benzaldehyde and
(f)
Interpretation:
The structure of the product expected when valine would react with aqueous Fmoc-![]() followed by neutralizing with
followed by neutralizing with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with aqueous Fmoc-
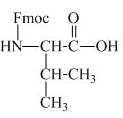
The structure of the product expected when proline would react with aqueous Fmoc-
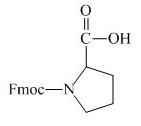
Explanation of Solution
The valine has an amine group. This group can be protected by the reaction of valine with aqueous Fmoc-
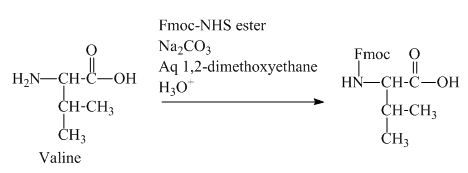
Figure 11
The proline has an amine group. This group can be protected by the reaction of proline with aqueous Fmoc-
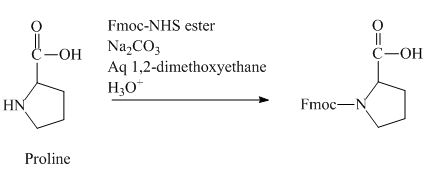
Figure 12
The structure of the product expected when valine would react with aqueous Fmoc-
The structure of the product expected when proline would react with aqueous Fmoc-
(g)
Interpretation:
The structure of the product expected when the given derivative of valine would react with ![]() butyl ester is to be predicted. The structure of the product expected when the given derivative of proline would react with
butyl ester is to be predicted. The structure of the product expected when the given derivative of proline would react with ![]() butyl ester is to be predicted.
butyl ester is to be predicted.
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with ![]() butyl ester is shown below.
butyl ester is shown below.
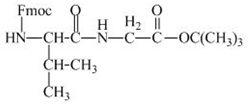
The structure of the product expected when the given derivative of proline would react with ![]() butyl ester is below.
butyl ester is below.
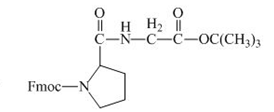
Explanation of Solution
The valine has an amine group. This group can be protected by the reaction of valine with aqueous Fmoc-![]() butyl ester. The corresponding chemical reaction is shown below.
butyl ester. The corresponding chemical reaction is shown below.
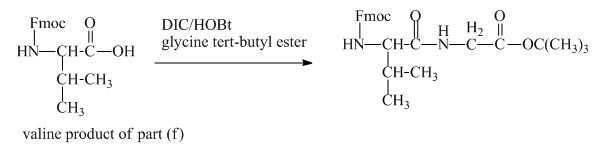
Figure 13
The carboxylic group of the derivative of proline can be protected with the help of reagent with ![]() butyl ester. The corresponding chemical reaction is shown below.
butyl ester. The corresponding chemical reaction is shown below.
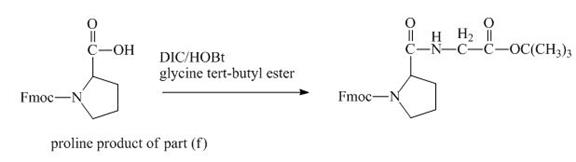
Figure 14
The structure of the product expected when the given derivative of valine would react with ![]() butyl ester is shown in Figure 13.
butyl ester is shown in Figure 13.
The structure of the product expected when the given derivative of proline would react with ![]() butyl ester is shown in Figure 14.
butyl ester is shown in Figure 14.
(h)
Interpretation:
The structure of the product expected when the given derivative of valine would react with anhydrous
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These functional groups can be protected by different reagent to synthesize peptide chains. The protecting groups can be removed by hydrolysis.
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with anhydrous
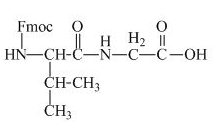
The structure of the product expected when the given derivative of proline would react with anhydrous
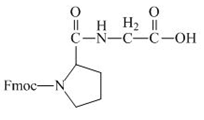
Explanation of Solution
The derivative of valine has an ester linkage in it. The acid,
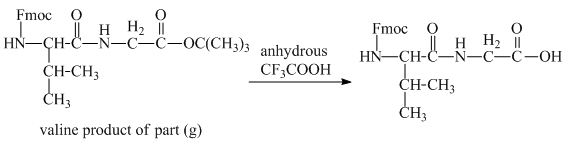
Figure 15
The derivative of proline has an ester linkage in it. The acid,
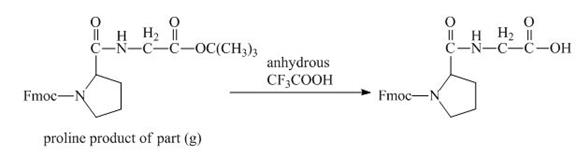
Figure 16
The structure of the product expected when the given derivative of valine would react with anhydrous
The structure of the product expected when the given derivative of proline would react with anhydrous
(i)
Interpretation:
The structure of the product expected when the given derivative of valine would react with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These functional groups can be protected by different reagent to synthesize peptide chains. The protecting groups can be removed by hydrolysis.
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with
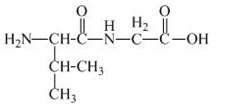
The structure of the product expected when the given derivative of proline would react with
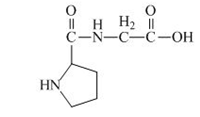
Explanation of Solution
The solution of
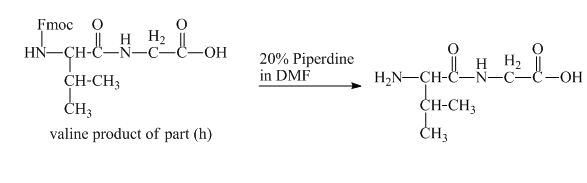
Figure 17
The solution of
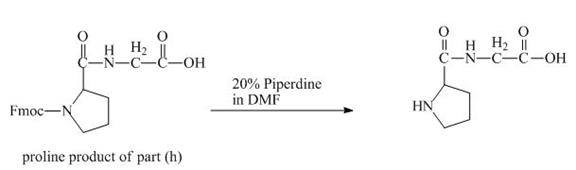
Figure 18
The structure of the product expected when the given derivative of valine would react with
The structure of the product expected when the given derivative of proline would react with
(j)
Interpretation:
The structure of the product expected when the given derivative of valine would react with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These functional groups can be protected by different reagent to synthesize peptide chains. The protecting groups can be removed by hydrolysis.
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with
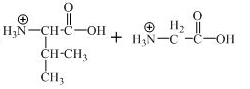
The structure of the product expected when the given derivative of proline would react with
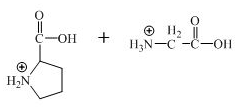
Explanation of Solution
The derivative of valine has amide linkage in it. This amide linkage can be hydrolysis by heating with a strong acid such as
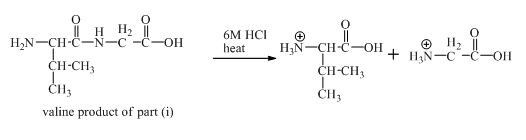
Figure 19
The derivative of valine has amide linkage in it. This amide linkage can be hydrolysis by heating with a strong acid such as
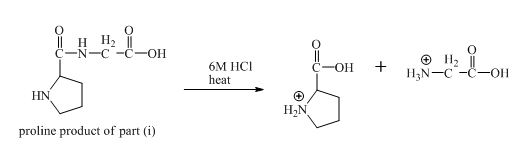
Figure 20
The structure of the product expected when the given derivative of valine would react with
The structure of the product expected when the given derivative of proline would react with
Want to see more full solutions like this?
Chapter 27 Solutions
Organic Chemistry
- (a) Draw a structural formula for the products formed when alanine is treated with the following reagents. (i) (Boc)20, NaOH (ii) CbzCI, NazCO3 (iii) 2-Methylpropene, p-TSOH (b) Enfuvirtide, a 36-amino-acid peptide drug, was manufactured at a multi-tonne per year scale. The manufacture relied on solid phase synthesis with Barlos resin as the solid support. Part of the solid phase synthesis is shown below. Write the structure of the product A and discuss the cleavage mechaniśm of the peptide from the resin. 1. Fmoc-Leu-OH, DIPEA 2. 20% piperidine in DMF 3. Fmoc-Glu('Bu)-OH, HBTU, HOBt 4. 20% piperidine in DMF 5. Fmoc-Leu-OH, HBTU, HOBT 6. 20% piperidine in DMF 7. Fmoc-Leu-OH, HBTU, HOBE 8. 20% piperidine in DMF 9. 10% HOAC in CH2CI2 Barlos resinarrow_forwardDraw the structure of the predominant form of a mixture of alanine, lysine, and aspartic acid at (i) pH 6; (ii) pH 11; (iii) pH 2.arrow_forwardConsider the following peptide : Phe – Glu – Ser – Met and Val – Trp – Cys – Leu. Do these peptides have different net charges at (a) pH 1? (b) pH 7? Indicate the charges at both pH valuesarrow_forward
- A biochemist completely digests a glycerophospholipid with a mixture of phospholipases A and D. HPLC and mass spectrometry analysis reveals the presence of an amino acid of 105,09 Da, a saturated fatty acid of 256.43 Da, and an omega-3 monounsaturated fatty acid of 282.45 Da. Which amino acid does the glycerophospholipid contain? O valine (C,H, NO,) O alanine (C, H,NO,) O scrine (C,H,NO,) O proline (C,H,NO,) Modify the phosphoglycerol backbone to draw the most likely structure of the lipid. Add the amino acid using the groups tool. Draw Rings Groups More Erase Selectarrow_forwardh) Specify the absolute (R/S) configuration of the amino group in structure IV. (i) If the substituents in structures I, IV and V were identical (all OH or all NH2), which structure would result in a meso compound? (j) If each hydroxy group for structures I, II and VI were replaced with another amino group, which compound would be made optically inactive?arrow_forwardAnswer the (b) part of the question 6 (a) A decapeptide has the following amino acid composition: Ala2 , Arg, Cys, Glu, Gly, Leu, Lys, Phe, Val Partial hydrolysis yields the following tripeptides: Cys-Glu-Leu + Gly-Arg-Cys + Leu-Ala-Ala+ Lys-Val-Phe + Val-Phe-Gly. Reaction of the decapeptide with 2,4-dinitrofluorobenzene yields 2,4-dinitrophenylysine. From the experimental data, deduce the primary structure of the decapeptide. (b) Suggest a scheme you will follow to synthesize the dipeptide Ala-Glyarrow_forward
- (a) (i) Write an equation showing the formation of a triacylglycerol (Molecule A) thatcontains equimolar amounts of myristoleic, capric and linolenic acids. Myristoleic acid: CH3(CH2)5CH=CH(CH2)7CO2HCapric acid: CH3(CH2)8CO2HLinolenic acid: CH3CH2(CH=CHCH2)3(CH2)6CO2H (ii) Draw the structure of the partially hydrogenated product of Molecule A.arrow_forwardDraw zwitterion forms of these amino acids. (a) Valine (b) Phenylalanine (c) Glutaminearrow_forwardThe peptide Proline-Serine-Alanine-Phenylalanine-Glutamine is present at pH 7. Draw the peptide and include stereochemistry.arrow_forward
- A tripeptide contains lysine (R= -(CH₂)4NH₂), phenylalanine (R= -CH₂C6H5) and glycine (R= -H). It was found that the tripeptide reacts with 2 moles of DNFB to give compound A. When compound A was treated with SOCI2, glycine was activated. When hydrolyzed, both NH₂ groups of lysine were found to be DNP-labelled. What is the structure of the tripeptide? Select one: H (CH₂)2NH2 CH NH CH H₂N O O H₂N H₂N H₂N C (CH2)4NH2 CH 010 (CH2)4NH2 CH C=O CH,Ph CH NH NH NH CH CH,Ph CH H 010 CH CH,Ph CH (CH2)4NH2 ΝΗ NH NH NH CH,Ph CH H CH COOH H CH COOH COOH COOHarrow_forwardExplain what is meant by the following :(i) peptide linkage(ii) pyranose structure of glucosearrow_forwardAspartame (Nutrasweet®) is a remarkably sweet-tasting dipeptide ester. Complete hydrolysis of aspartame gives phenylalanine, aspartic acid, and methanol. Mild incubation with carboxypeptidase has no effect on aspartame. Treatment ofaspartame with phenyl isothiocyanate, followed by mild hydrolysis, gives the phenylthiohydantoin of aspartic acid.Propose a structure for aspartame.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
