
Concept explainers
Interpretation:
The physical states that correspond to the pictorial representation are to be stated.
Concept introduction:
A substance which has some mass and occupies space is termed as matter. It exists in three physical states that are solid, liquid and gas. The force of attraction between atoms determines the
Answer to Problem 1CE
The illustration A, B, C represents the solid state, liquid state and gaseous state respectively.
Explanation of Solution
The given figure is shown below.
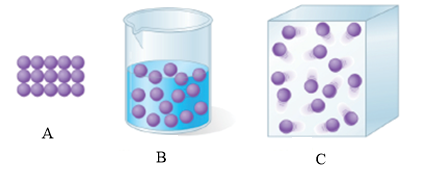
Figure 1
Illustration A is a solid state as the force of attraction between atoms is very strong and the atoms are in a definite shape. On the other hand, illustration B is a liquid state as the atoms are far from each other because the force of attraction between atoms is less. The illustration C represents the gaseous state as atoms are very far from each other because the force of attraction between atoms is negligible.
Illustration A, B, C represents solid state, liquid state and gaseous state respectively.
Want to see more full solutions like this?
Chapter 3 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Classify the substances represented by the following models as homoatomic or heteroatomic molecules.arrow_forwardWrite a brief description of the relationships among each of the following groups of terms or phrases. Answers to the Concept-Linking Exercises are given at the end of the chapter. Homogeneous, heterogeneous, pure substance, mixturearrow_forwardSeawater is composed of salt, sand, and water. Is seawatera heterogeneous or homogeneous mixture? Explain.arrow_forward
- Using spheres to represent individual atoms, sketch particulate illustrations of a substance as it is heated from the solid to the liquid and to the gaseous state.arrow_forwardIn the left box, draw a particulate-level illustration of a substance in the gaseous state. Model the particles as spheres, which can be simple circles. Assume that the box represents a tiny, closed container that holds the particles. In the right box, draw a particulate-level illustration of the same substance after it cools and becomes a liquidarrow_forwardchocolate candy compound and contituent elementarrow_forward
- Which of the following would be considered a heterogeneous mixture?arrow_forwardIndicate whether the following changes of physical state require heating or cooling. (a) gas to liquid (b)liquid to solidarrow_forwardNot sure what I am supposed to put in the blanks of "gas", "liquid", and "solid" and the rest of the blanksarrow_forward
- Explain the following changes in states of matter. Write one example for each. (A) Condensation (B) Sublimationarrow_forwardWater and laundry detergent are mixed together, with detergent molecules dispersing uniformly throughout the liquid. This is an example of a A) mixture of elements B) heterogeneous mixture C) homogeneous mixture (solution) D) molecular compoundarrow_forwardWhich of the following statements is FALSE according to the kinetic molecular theory? O Matter is composed of particles (atoms, molecules or ions). O Particles are in constant random motion and therefore possess kinetic energy. O Kinetic energy will increase as the temperature is increased. Particle interact through attractions and repulsions observed as potential energy. Particles transfer energy through temporary covalent bonding.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER




