
Concepts of Biology
1st Edition
ISBN: 9781938168116
Author: Samantha Fowler, Rebecca Roush, James Wise
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3, Problem 3ACQ
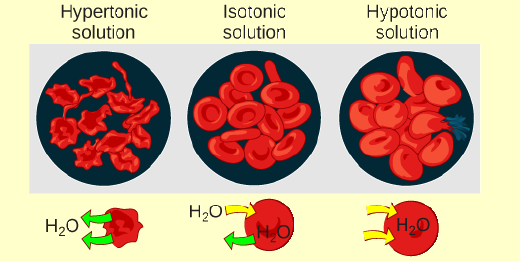
Figure 3.22 A doctor injects a patient with what he thinks is isotonic saline solution. The patient dies, and autopsy reveals that many red blood cells have been destroyed. Do you think the solution the doctor injected was really isotonic?

Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
If a doctor injected a patient with what was labeled as an isotonic saline solution, but then the patient died, and an autopsy revealed that several of the patient's red blood cells had burst, such as in the image on the right, would it be true that the injected solution was really isotonic? Why or why not?
In the diagram below, which situation is when the red blood cell is exposed to hypertonic solution? What is happening to the
red blood cells in that situation?
(a)
(b)
(c)
O c: hemolysis
O c; crenation
a; hemolysis
O a; crenation
Intravenous solutions must be prepared so that they are isotonic to red blood cells. A 0.9% salt solution is isotonic. What will happen to a red blood cell placed in a 0.6% salt solution?
Chapter 3 Solutions
Concepts of Biology
Ch. 3 - Figure 3.7 What structures does a plant cell have...Ch. 3 - Figure 3.13 Why does the cis face of the Golgi not...Ch. 3 - Figure 3.22 A doctor injects a patient with what...Ch. 3 - When viewing a specimen through a light...Ch. 3 - The ____________ is the basic unit of life. a....Ch. 3 - Which of these do all prokaryotes and eukaryotes...Ch. 3 - A typical prokaryotic cell ________ compared to a...Ch. 3 - Which of the following is found both in eukaryotic...Ch. 3 - Which of the following is not a component of the...Ch. 3 - Which plasma membrane component can be either...
Ch. 3 - The tails of the phospholipids of the plasma...Ch. 3 - Water moves via osmosis _______. a. throughout the...Ch. 3 - The principal force driving movement in diffusion...Ch. 3 - Active transport must function continuously...Ch. 3 - What are the advantages and disadvantages of...Ch. 3 - Describe the structures that are characteristic of...Ch. 3 - In the context of cell biology, what do we mean by...Ch. 3 - Why is it advantageous for the cell membrane to be...Ch. 3 - Why does osmosis occur?Ch. 3 - Where does the cell get energy for active...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The great astronomer Edwin Hubble discovered that all distant galaxies are receding from our Milky Way Galaxy w...
College Physics
1. Which trophic level(s) must be present to sustain any ecosystem?
a. producers only
b. producers and consume...
Biology: Life on Earth (11th Edition)
Compare and contrast aerobic respiration, anaerobic respiration, and fermentation.
Microbiology with Diseases by Body System (4th Edition)
CAUTION Why does the presence of extinct forms and transitional features in the fossil record support the patte...
Biological Science
1. What are the main organs of the skeletal system?
Human Anatomy & Physiology
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Figure 5.12 A doctor injects a patient with what the doctor thinks is an isotonic saline solution. The patient dies, and an autopsy reveals that many red blood cells have been destroyed. Do you think the solution the doctor injected was really isotonic?arrow_forwardIn the following diagram, identify the type of solution (hypertonic, hypotonic, or isotonic) in which each red blood cell is immersed.arrow_forwardIsotonic saline and 5% dextrose in water are solutions considered isotonic to human blood. What effect on red blood cells would you expect if a patient were given these fluids intravenously? A solution of 10% dextrose in water is hypertonic to human blood.arrow_forward
- A patient has had a serious accident and lost a lot of blood. In an attempt to replenish body fluids, a large amount of distilled water was transferred directly into one of his veins. What will be the most probable result of this transfusion? It will have serious, perhaps fatal, consequences because the red blood cells will be hypertonic relative to the body fluids and the cells will burst. It will have serious, perhaps fatal, consequences because the red blood cells will be hypotonic relative to the body fluids and the cells will shrivel. It will have no unfavorable effect as long as the water is free of viruses and bacteria. It will have no serious effect because the kidneys would quickly eliminate the excess water. It will have serious, perhaps fatal, consequences because there will be too much fluid for the heart to pump.arrow_forwardWhich answer is correct (a or b) for the question about why your hands and feet get wrinkled after staying in the bath for too long. a) the water and dissolved particles are drawn out of your skin by diffusion into the large body of water around you. b) the water is actually drawn into your skin by osmosis. explain why (a or b) is correct.arrow_forwardInflammatory responses are associated with lots of swelling, where fluids rush into cells and surrounding tissues. To help quickly relieve swelling, what type of IV solution could a doctor administer to her patient? hypotonic hypertonic isotonicarrow_forward
- Why is it important to know the isotonic point of human cells when administering an IV? Explain what would happen to the red blood cells if an IV solution were hypotonic or hypertonic.arrow_forwardIsotonic, hypotonic, and hypertonic IV (intravenous) fluid solutions are widely used in the healthcare setting. As a possible health care professional, you must know how each of the solutions work on the body and why they are given. a) When does each type of solution must be applied to a patient? b) What does each type of solution do the cells? Isotonic IV fluid solution Hypotonic IV fluid solution Hypertonic IV fluid solution|arrow_forwardYour friend notes that Gatorade has electrolytes like sodium and potassium chloride. He figures that if a few electrolytes are good for you, then a lot must be even better. Following this logic, he adds nearly a tablespoonful of salt to his Gatorade. What kind of solution is his Gatorade now (hypotonic, isotonic, or hypertonic)? What will likely happen to his cells if he drinks this liquid?arrow_forward
- In medicine, why is it important to administer only isotonic intravenous solutions to patients? Example of isotonic IV fluids are normal saline and lactated Ringer’s solution.arrow_forwardIf a red blood cell was placed in a 50mOSM solution, the cell would:arrow_forwardMany old time folk remedies made use of the priciples of diffusion and osmosis. Suppose you had a sliver of wood or another small foreign object embedded under your skin. It is slightly swollen and hurts. Your grandma or older neighbor tells you to soak the affected part in salt water. Using your knowledge of diffusion/osmosis, how do you think this would affect the injury? Will it help you get the sliver out?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax
Necrosis vs. Apoptosis: Cell Death; Author: AMBOSS: Medical Knowledge Distilled;https://www.youtube.com/watch?v=zFrBwGfOQs0;License: Standard Youtube License