
Concept explainers
5-114 Carbon dioxide gas, saturated with water vapor, can be produced by the addition of aqueous acid to calcium carbonate based on the following balanced net ionic equation:
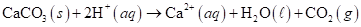
(a) How many moles of wet CO (g), collected at 60.°C and 774 torr total pressure, are produced by the complete reaction of 10.0 g of CaCO3 with excess acid?
(b) What volume does this wet CO2 occupy?
(c) What volume would the CO2 occupy at 774 torr if a desiccant (a chemical drying agent) were added to remove the water? The vapor pressure of water at 60.°C is 149.4 mm Hg.
(a)
Interpretation:
The number of moles of wet
Concept Introduction:
Moles of wet
Answer to Problem 5.114P
The number of moles of carbon dioxide produced is
Explanation of Solution
The weight of calcium carbonate is
The temperature is
The pressure is
The molecular weight of calcium carbonate is calculated below:
The number of moles in
The balanced chemical equation is below:
From the above equation,
Therefore, the number of moles of carbon dioxide produced is
(b)
Interpretation:
The volume of wet
Concept Introduction:
To determine the volume of wet
The relationship between pressure, volume, and temperature, under two sets of conditions, is given by following equation.
Answer to Problem 5.114P
The value of carbon dioxide produced is
Explanation of Solution
The weight of calcium carbonate is
The temperature is.
The pressure is
The molecular weight of calcium carbonate is calculated below:
The number of moles in
The balanced chemical equation is below:
From the above equation:
Therefore, the number of moles of carbon dioxide produced is
We know that at STP, 1 mole of any gas occupies
Therefore,
Therefore, the volume of
At STP, the value of
Now, convert the pressure from
We know that,
Therefore, the pressure is calculated below:
Now, convert temperature from
We know that,
Therefore,
The relationship between pressure, volume, and temperature, under two sets of conditions, is given by following equation:
Upon rearranging, we get,
By substituting the values of
Hence the value of carbon dioxide produced is
(c)
Interpretation:
The volume of wet
Concept Introduction:
To determine the volume of wet
The relationship between pressure, volume, and temperature, under two sets of conditions, is given by following equation.
Answer to Problem 5.114P
Explanation of Solution
The weight of calcium carbonate is
The temperature is
The pressure is
The vapor pressure of water at
The molecular weight of calcium carbonate is calculated below:
The number of moles in
The balanced chemical equation is below:
From the above equation:
Therefore, the number of moles of carbon dioxide produced is
We know that at STP, 1 mole of any gas occupies
Therefore,
Therefore, the volume of
At STP, the value of
The pressure is
Now, convert the pressure from
We know that,
Therefore, the pressure is calculated below,
The vapor pressure of water at
Therefore, the pressure is calculated below,
Now, convert temperature from°C to K.
We know that,
Therefore,
The relationship between pressure, volume, and temperature, under two sets of conditions, is given by following equation,
Upon rearranging, we get,
By substituting the values of
Hence the value of carbon dioxide produced is
Want to see more full solutions like this?
Chapter 5 Solutions
Introduction to General, Organic and Biochemistry
- What mass of KO2 is required to remove 90.0% of the CO2 from a sample of 1.00 L of exhaled air (37°C, 1.00 atm) containing 5.00 mole percent CO2?arrow_forward6-78 (Chemical Connections 6A) Oxides of nitrogen (NO, NO2,N2O3) are also responsible for acid rain. Which acids can be formed from these nitrogen oxides?arrow_forwardHow does hydraulic fracturing differ from previously used techniques for the recovery of natural gas from the earth?arrow_forward
- What possible uses exist for the natural gas liquids that are removed from natural gas during its processing?arrow_forwardHow many liters of HCI gas, measured at 30.0 C and 745 torr, are required to prepare 1.25 L of a 3.20-M solution of hydrochloric acid?arrow_forwardGiven that a sample of air is made up of nitrogen, oxygen, and argon in the mole fractions 0.78 N2, 0.21 O2, and 0.010 Ar, what is the density of air at standard temperature and pressure?arrow_forward
- (a) A rigid tank contains 1.60 moles of helium, which can be treated as an ideal gas, at a pressure of 28.0 atm. While the tank and gas maintain a constant volume and temperature, a number of moles are removed from the tank, reducing the pressure to 5.00 atm. How many moles are removed? mol (b) What If? In a separate experiment beginning from the same initial conditions, including a temperature T, of 25.0°C, half the number of moles found in part (a) are withdrawn while the temperature is allowed to vary and the pressure undergoes the same change from 28.0 atm to 5.00 atm. What is the final temperature (in °C) of the gas? °Carrow_forwardSulfur dioxide reacts with oxygen in the presence of plati- num to give sulfur trioxide: 2 SO2(g) + O2(g) → 2 SO3(g) Suppose that at one stage in the reaction, 26.0 mol SO2, 83.0 mol O2, and 17.0 mol SO3 are present in the reaction vessel at a total pressure of 0.950 atm. Calculate the mole fraction of SO3 and its partial pressure.arrow_forwardCalculate the partial pressure of O2 produced from 1.80 L of 0.576 M N2O5 solution at 45 ∘C over a period of 20.8 h if the gas is collected in a 10.3-Lcontainer. (Assume that the products do not dissolve in chloroform.)arrow_forward
- The estimated average concentration of NO2 in air in theUnited States in 2006 was 0.016 ppm. (a) Calculate thepartial pressure of the NO2 in a sample of this air whenthe atmospheric pressure is 755 torr (99.1 kPa). (b) Howmany molecules of NO2 are present under these conditionsat 20 °C in a room that measures 15 x 14 x8 ft?arrow_forwardIf 14.5g of calcium carbonate decomposes via the following reaction, how many liters of carbon dioxide at a temperature of 303K and 1.02atm will be produced? CaCO3(s)−→−heatCaO(s)+CO2(g) Use R=0.08206L atmmol K for the ideal gas constant. Your answer should have three significant figures.arrow_forward5. Calculate the volume of oxygen consumed at SATP (25 °C, 100kPa) by the combustion of 10.4 kg of propane, C3H8. (arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





