
Concept explainers
(a)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
![]()
Explanation of Solution
The
![]()
Figure 1
The electron dot structure of
(b)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the symbol of an element is written.
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
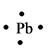
Explanation of Solution
Lead belongs to the group
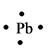
Figure 2
The electron dot structure of
(c)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the symbol of an element is written.
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
![]()
Explanation of Solution
Selenium belongs to the group
![]()
Figure 3
The electron dot structure of
(d)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the symbol of an element is written.
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
![]()
Explanation of Solution
Neon belongs to the group
![]()
Figure 4
The electron dot structure of
(e)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the symbol of an element is written.
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
![]()
Explanation of Solution
Cesium belongs to the group
![]()
Figure 5
The electron dot structure of
(f)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the symbol of an element is written.
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
![]()
Explanation of Solution
Gallium belongs to the group
![]()
Figure 6
The electron dot structure of
(g)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the symbol of an element is written.
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
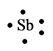
Explanation of Solution
Antimony belongs to the group
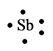
Figure 7
The electron dot structure of
(h)
Interpretation:
The electron dot structure for the atom of
Concept introduction:
The electron dot formula is used for the identification of valence electrons present in an atom. It shows the symbol of an atom with its valence electron around it. It is also called as Lewis dot structure. The valence electrons around an atom are shown by dots. One dot represents one valence electron.
The rules to draw electron dot formula are given below.
• First, the symbol of an element is written.
• With the help of group number, the number of valence electrons is determined.
• The valance electrons are drawn around the atom by dots.
Answer to Problem 62E
The electron dot structure for the atom of
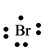
Explanation of Solution
Bromine belongs to the group
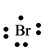
Figure 8
The electron dot structure of
Want to see more full solutions like this?
Chapter 5 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- How many inner, outer, and valence electrons are present in an atom of each of the following elements?(a) O(b) Sn(c) Ca(d) Fe(e) Searrow_forwardHow many inner, outer, and valence electrons are present in an atom of each of the following elements? (a) Br (b) Cs (c) Cr (d) Sr (e) Farrow_forwardWhat are the chemical formulas for ( a ) disulfur dioxide and ( b ) iodine pentafluoride ?arrow_forward
- Draw the electron dot formula for each of the following elements: (a) Si (b)Xearrow_forwardHow many inner, outer, and valence electrons are present inan atom of each of the following elements?(a) Br(b) Cs(c) Cr(d) Sr(e) Farrow_forwardWhich of the following elements is expected to have the highest SECOND IONIZATION ENERGY: (A) Be (B) Ca (C) Mg (D) Naarrow_forward
- Using Lewis electron-dot symbols to depict the monatomic ions formed from each of the following reactants, predict the formula of the compound the ions produce.(Type your answer using the format CO2 for CO2.) (a) O and Ca (b) N and Mg (c) Br and Li (d) K and Parrow_forwardWhich of these elements is most likely to form ions with a 2+charge?(a) Li (b) Ca (c) O (d) P (e) Clarrow_forwardWrite the ionic charge for each of the following ions as predicted from the group number in the periodic table. (a) Cs ion (b) Ga ion (c) O ion (d) I ionarrow_forward
- In each of the following pairs, which has the larger radius? (a) Li atom or Li ion (b) Mg atom or Mg ion (c) F atom or F ion (d) O atom or O ionarrow_forwardAssume that you encounter the following phrases in your reading. What is the chemical formula for each substance mentioned? (a) Sodium Hydrogen carbonate is used as a deodorant. (b) Calcium hypochlorite is used in some bleaching solutions. (c) Hydrogen cyanide is a very poisonous gas. (d) Magnesium hydroxide is used as a cathartic. (e) Tin (II) fluoride has been used as a fluoride additive in toothpastes. (f) When cadmium sulfide is treated with sulfuric acid, fumes of hydrogen sulfide are given off.arrow_forwardWhich of the following compounds has both ionic and covalent bond? (a) H2O (b) NH4Cl (c) NaCl (d) HClarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





