
Concept explainers
Propose a mechanism for each of the following reactions:
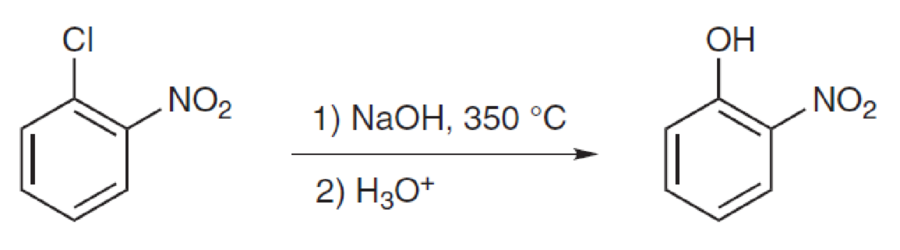
Interpretation:
Mechanism for the given transformation has to be proposed.
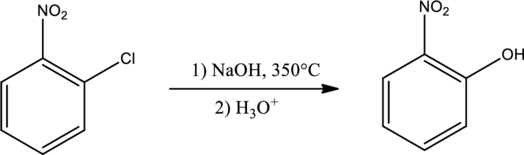
Explanation of Solution
Given reaction is,
Mechanism:
Looking into the reaction it is found that the nucleophile is a strong one namely hydroxide and the reactant has all the three important criteria to undergo
First step is the attack of nucleophile on the carbon atom that bears that chlorine atom which is a leaving group resulting in formation of Meisenheimer complex.
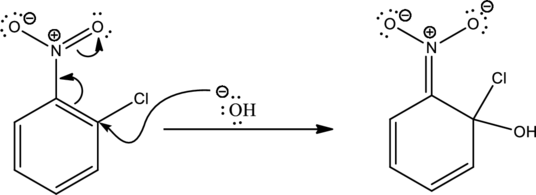
The formed Meisenheimer complex has the following resonance structures.
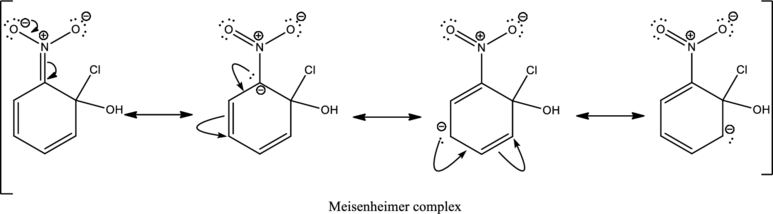
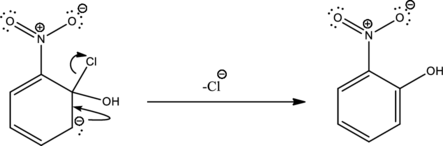
Second step is the expelling of the leaving group to give the product.
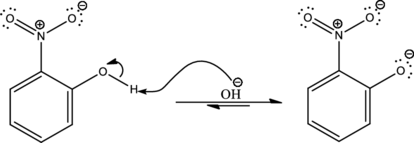
As the given condition is basic, deprotonation takes place.
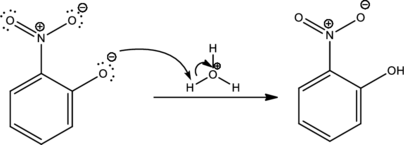
Under acidic condition the formed ion is protonated to give the product. This is the reason for using acid in order to complete the reaction.
Complete mechanism can be given as shown below,
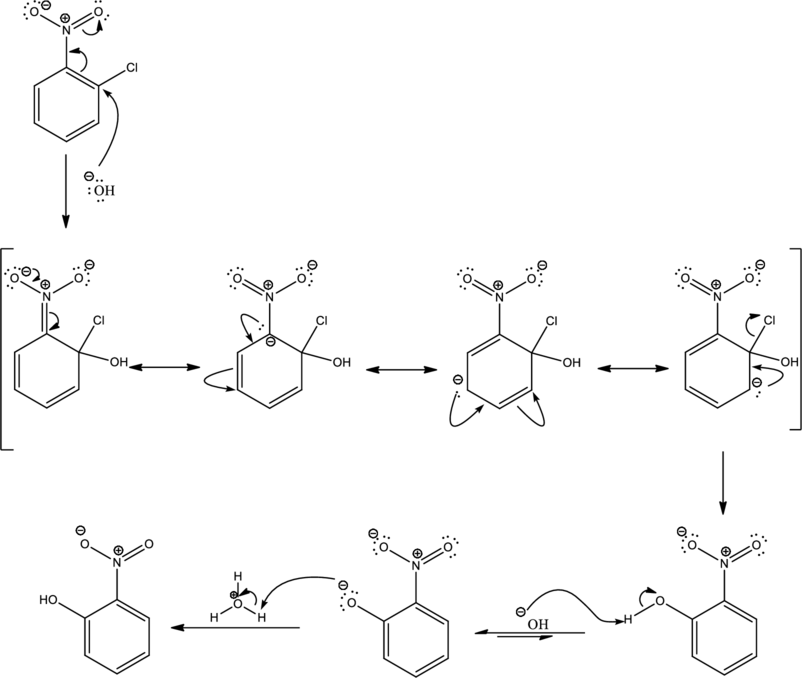
Mechanism for the given transformation is proposed.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
Basic Chemistry (5th Edition)
Living by Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
Living By Chemistry: First Edition Textbook
Essential Organic Chemistry (3rd Edition)
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardIsolated from marine algae, prelaureatin is thought to be biosynthesized from laurediol by the following route. Propose a mechanism.arrow_forwardThe Meerwein-Ponndorf-Verley reaction involves reduction of a ketone by treatment with an excess of aluminum triisopropoxide, [(CH3)2CHO]3Al. The mechanism of the process is closely related to the Cannizzaro reaction in that a hydride ion acts as a leaving group. Propose a mechanism.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

