
(a)
To determine: The method of conversion of
Interpretation: The method of conversion of
Concept introduction: NBS is N-bromosuccinimide. It is used as an alternative for
(a)
Answer to Problem 6.30SP
The conversion of
Explanation of Solution
The conversion of
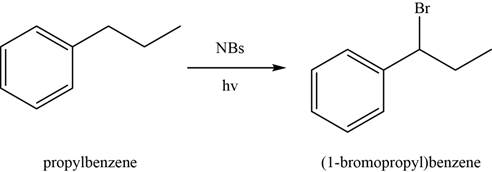
Figure 1
In the above reaction,
(b)
To determine: The method of conversion of
Interpretation: The method of conversion of
Concept introduction: NBS is N-bromosuccinimide. It is used as an alternative for
(b)
Answer to Problem 6.30SP
The conversion of
Explanation of Solution
In first step,
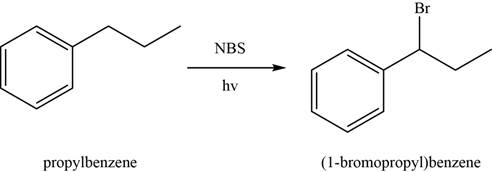
Figure 2
In the second step,
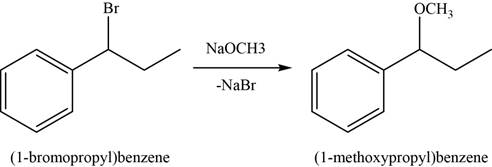
Figure 3
(c)
To determine: The method of conversion of
Interpretation: The method of conversion of
Concept introduction: NBS is N-bromosuccinimide. It is used as an alternative for
(c)
Answer to Problem 6.30SP
The conversion of
Explanation of Solution
In first step,
In the second step,
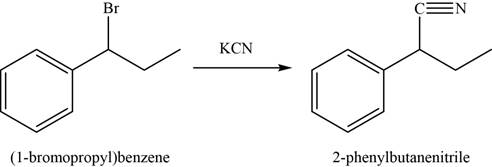
Figure 4
Want to see more full solutions like this?
Chapter 6 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
- Give a detailed reaction mechanism for the reaction expected to occur when 2-bromo-2-methylpentane is heated with sodium methoxide. Draw clear structural formulas of all relevant species and use curved arrows to represent electron flow. Also indicate which step is likely to be rate-determining. The answer you sent before will be used for this question.arrow_forwardGive the products of the following substitution reactions. For every reaction, show electron pairs on both nucleophile and leaving group.arrow_forwardProvide the reagents for the short syntheses (about 3-4 steps).arrow_forward
- Complete the synthesis of the following compound from the starting material given (2- oxocyclohexane-1-carbaldehyde). Over/next to the reaction arrows, list out all reagents/solvents used. Draw intermediate structures in the boxes. You may use as many steps as you think necessary. You do NOT have to draw the mechanisms.arrow_forwardList at least three (3) physical or spectroscopic methods that can be used to differentiate between benzophenone and benzhydrol. Explain how you would differentiate them. What are the most likely impurities to be found in the product of benzhydrol synthesis? How will they affect performance? How can its presence in the final product be reduced?arrow_forwardAcyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the tetrahedral intermediate as it is first formed in the following reaction. You do not have to consider stereochemistry. Include all valence lone pairs in your answer. Do not include counter-ions, e.g., Na+, I-, in your answer. In cases where there is more than one answer, just draw one.arrow_forward
- IF esize this compound by the → t-butyl ethyl ether Show the steps necessary to synthesize this compound by a rignard reaction. Start with an alkyl halide; after that you can add any organic or inorganic compound. → 1-hexanol Consider the following compounds:arrow_forwardComplete the synthesis of ethyl (E)-2,2,4-trimethyl-3-oxo-5-phenylpent-4-enoate from the starting material given (ethyl propionate). You must list out all reagents/solvents next to the reaction arrows and draw the intermediate structures in the boxes. I am giving you one restriction. You are not allowed to introduce any enolates as reagents next to the reaction arrows.arrow_forwardThe question is: "Draw the curved arrow mechanism for the reaction between pentan-2-one and (CH3)3O– in t-butanol to form an enolate. Draw all electrons and charges on both resonance structures. Then answer the question about the reaction." I got the initial arrows correct, but am not entirely sure what the carbanion intermediate would look like and then what the curved arrows would be to convert it to its final oxanion formarrow_forward
- Complete the synthesis of ethyl (E)-2,2,4-trimethyl-3-oxo-5-phenylpent-4-enoate from the starting material given (ethyl propionate). You must list out all reagents/solvents next to the reaction arrows and draw the intermediate structures in the boxes. I am giving you one restriction. You are not allowed to introduce any enolates as reagents next to the reaction arrows. orff ionarrow_forward3. Perform a retrosynthetic analysis on the following alkene. Show both ways of making it from alkyl halides and carbonyl compounds. After you have shown both syntheses, suggest which of the two would be a better one. Show both starting material combinations with your model kit and share pictures. Using the model kit will help you with getting the answer.arrow_forwardHow would you synthesize the following compounds from cyclohexanone using reagents from the table? Use letters from the table to list reagents in the order used (first at the left). Reagents a 1. CH3MgBr / dry ether 2 H3O+ e 1. OsO4 i 1. BH3/THF 2. NaHSO3/H₂O 2. H₂O₂ / NaOH b 1. C6H5 MgBr / dry ether f 1. NaBH4 j Mg / dry ether 2 H3O+ 2. H3O+ C PBг3 g HOCH2CH2OH/HCI k H2/Pd d m-chloroperbenzoic h H3O+ / heat 1 CrO3 / H3O+ acid a) 2-phenylcyclohexanone b) cyclohexylbenzene m cyclohexanonearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

