
Concept explainers
Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book.
Drawing Haworth Structures of Sugars Draw Haworth structures for the two possible isomers of D-altrose (Figure 7.2) and D-psicose (Figure 7.3).
Interpretation: The Haworth structures for two possible isomers of
Concept introduction: The simplest hydrolyzed form that is obtained from the carbohydrates is known as a monosaccharide.
Haworth projection is a method that is used to provide the three-dimensional cyclic form of a monosaccharide. This method shows the monosaccharide in the form of a hemiacetal ring or hemiketal ring.
Carbon atom which behaves as a stereocenter or a chiral center in the cyclic ring form of the hemiacetal or hemiketal compound is known as anomeric carbon.
Answer to Problem 1P
The Haworth structures for two possible isomers of
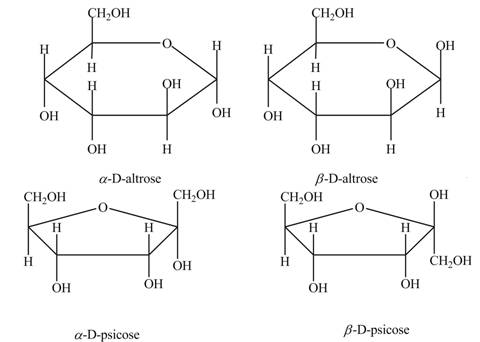
Explanation of Solution
The anomeric carbon of a monosaccharide unit always possesses two types of isomers. One is
The molecular formula of
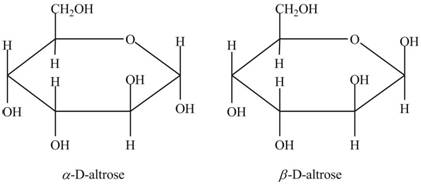
Figure 1
In the Haworth projection of
The molecular formula of
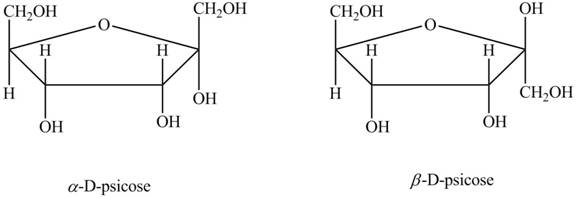
Figure 2
In the Haworth projection of
Want to see more full solutions like this?
Chapter 7 Solutions
Biochemistry
- Answers to all problems are at the end of this book.. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Drawing the Fischer Projection of a Simple Sugar Draw a Fischer projection structure for L-sorbose (D-sorbose is shown in Figure 7.3).arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. (Historical Context) The Third Person of the -Helix Publication Who was Herman Branson? What was his role in the elucidation of the structure of the or-helix'.' Did he receive sufficient credit and recognition for his contributions? And how did the rest of his career unfold? Do a Google search on Herman Branson to learn about his life, and read the article by David Eisenberg under Further Reading. You may also wish to examine the original paper by Pauling, Corey, and Branson, as well as the following Web site: http://www.pirns. org/sitelmisclclassicsl..shtml Pauling, L., Corey, R. B., and Branson, H. R., 1951. The structure of proteins: two hydrogen-bonded helical configurations of the polypeptide chain. Proceedings of the National Academy of Sciences, USA 37:235-240.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Interpreting Kinetics Experiments from Graphical Patterns The following graphical patterns obtained from kinetic experiments have several possible interpretations depending on the nature of the experiment and the variables being plotted. Give at least two possibilities for each.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. (Research Problem) The Nature and Roles of Linear Motifs in Proteins In addition to domains and modules, there are other significant sequence patterns in proteins—known as linear motifs—that are associated with a particular function. Consult the biochemical literature to answer the following questions: 1. What are linear motifs? 2. How are they different from domains?. 3. What are their functions? 4. How can they be characterized? 5. There are several papers that are good starting points for this problem. Neduva, V., and Russell, R., 2005. Linear motifs: evolutionary interaction switches. FEBS Letters 579:3342-3345. Gibson, T., 2009. Cell regulation: determined to signal discrete cooperation. Trends in Biochemical Sciences 34:471-482. Diella, K. Haslam, N., Chica., C. et aL, 2009. Understanding eukaryotic linear motifs and their role in cell signaling and regulation. Frontiers of Bioscience 13:6580-6603.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Oligonucleotide Structure Draw the chemical structure of pACG.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Writing Dissociation Equations for Amino Acids Write equations fur the ionic dissociations of alanine, glutamate, histidine, lysine, and phenylalanine.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Proteins and nucleic acids are informational macromolecules. What are the two minimal criteria for a linear informational polymer?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Draw all the possible isomers of threonine and assign (R,S) nomenclature to each.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Drawing Fischer Projection Formulas for Amino Acids Without consulting chapter figures, draw Fischer projection formulas for glycine, aspartate, leucine, isoleucine, methionine, and threonine.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. To fully appreciate the elements of secondary structure in proteins, it is useful to have a practical sense of their structures. On a piece of paper, draw a simple but large zigzag pattern to represent a -strand. Then fill in the structure, drawing the locations of the moms of the chain on this zigzag pattern. Then draw a simple, large coil on a piece of paper to represent an -helix. Then fill in the structure, drawing the backbone atoms in the correction locations along the coil and indicating the locations of the R groups in your drawing.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Use examples from the ActiveModel for Human GaleLtin-1 to describe the hydrophobic effect.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. The dissociation constant for a particular protein dimer is 1 micromolar. Calculate the free energy difference for the monomer-to-dimer transition.arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
