
(a)
Interpretation:
The energy cost of a
Concept introduction:
In a cyclohexane ring, the steric interactions that takes place between an axial substituent located on the carbon atom 1 and the hydrogen atoms located on carbon 3 and 5 is known as 1, 3 diaxial interaction. In the cyclohexane derivatives, each 1, 3-diaxial interaction between the methyl and hydrogen increases the enthalpy of the ring by
Answer to Problem 7.72AP
The energy cost of a
Explanation of Solution
The two conformations of cyclohexane are given below.
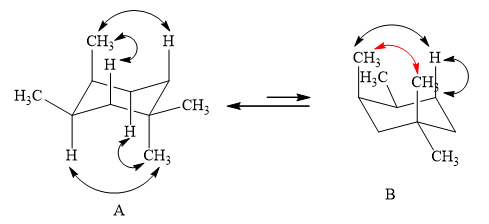
Figure 1
In the above shown figure, in conformation
The formula to calculate
The
Substitute the values of energy and
Therefore, the energy between two methyl groups is
The energy between two methyl groups is
(b)
Interpretation:
The value of
Concept introduction:
In a cyclohexane ring, the steric interactions that takes place between an axial substituent located on the carbon atom 1 and the hydrogen atoms located on carbon 3 and 5 is known as 1, 3 diaxial interaction. In the cyclohexane derivatives, each 1, 3-diaxial interaction between the methyl and hydrogen increases the enthalpy of the ring by
Answer to Problem 7.72AP
The
Explanation of Solution
The two conformations of cyclohexane are given below.
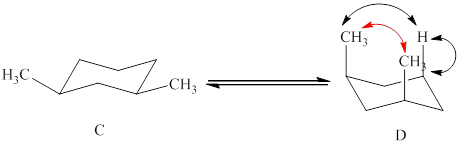
Figure 2
In the above shown figure, in conformation
The formula to calculate
The energy for every methyl-hydrogen
The energy for methyl-methyl
Substitute the values of energy into the above equation.
The value of
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





