
Concept explainers
(a)
Interpretation: The order of stability is to be interpreted for the given
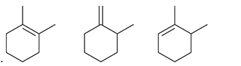
Concept introduction:
(b)
Interpretation: The order of stability is to be interpreted for the given alkenes:
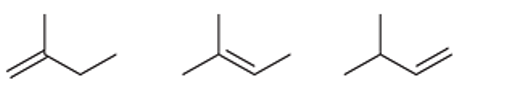
Concept introduction:
Unsaturated hydrocarbons with at least one double bond between the carbon atoms are known as alkenes. The presence of pi bonds in these molecules makes them more reactive compared to saturated hydrocarbons; alkanes. The stability of alkenes depends on their substitution.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
- Gibbs free energy differences between axial-substituted and equatorial-substituted chair conformations of cyclohexane were given in Table 2.4. (a) Calculate the ratio of equatorial to axial tert-butylcyclohexane at 25C. (b) Explain why the conformational equilibria for methyl, ethyl, and isopropyl substituents are comparable but the conformational equilibrium for tert-butylcyclohexane lies considerably farther toward the equatorial conformation.arrow_forwardMatch each alkene to its heat of hydrogenation. Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene, 2-methylbut-2-ene ΔH° (hydrogenation) kJ/mol: –119, –127, –112arrow_forwardWhich one alkene will exhibit cis-trans isomerism? A B OC OA u B Darrow_forward
- 2. Explain the selectivity of the following reaction, which produces a single product despite both alkene carbons being equally substituted. H3C CF3 HBr CF3 H3C CF3 H3C Br- H3C only product -H CF3arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. CH3 HCI CH3 CHCCH, ? + Či CH3 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one.arrow_forwardH,C -CH3 H,C -CH3 CH3 H,C CH structure 1 structure 2 Which is the more stable alkene? Explain your answer.arrow_forward
- Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to give alkyl halides having molecular formulaC6H13Cl. A yields five constitutional isomers. B yields four constitutionalisomers. C yields two constitutional isomers. D yields threeconstitutional isomers, two of which possess stereogenic centers. Eyields three constitutional isomers, only one of which possesses astereogenic center. Identify the structures of A–E.arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + + H₂O **** H₂S04 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH3 CHCCH3 | | OH CH3 +1arrow_forwardCH3CH2CH2 H CH3CH2CH2 CH3 H2C=CHCH,CH2CH2CH3 CH3 B Which of the alkenes above is the least stable (highest in energy)? Which is the most stable (lowest in energy)?arrow_forward
- Draw the alternative chair conformations for the product formed by the addition of bromine to 4-tert-butylcyclohexene. The Gibbs free energy differences between equatorial and axial substituents on a cyclohexane ring are 21 kJ (4.9 kcal)/mol for tert-butyl and 2.0–2.6 kJ (0.48–0.62 kcal)/mol for bromine. Estimate the relative percentages of the alternative chair conformations you drew in the first part of this problemarrow_forward1. Illustrate details mechanism of reactions for bromination of the following alkane. State each of the steps involves. CH3 Br2 CH `CH3 hv, 25°C H3C 2. Draw the two chair conformations of cis-1-chloro-4-methylcyclohexane. Which of the conformers is more stable?arrow_forwardName the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H CH3 H3C C=C H2C=CHCHÇH H ČH3arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
