
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.7, Problem 15P
Place a double bond in the carbon skeleton shown so as to represent
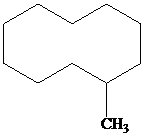
a)
b)
c)
d)
e)
f)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
6. What is the correct IUPÁC name of the following compound?
A) (2S, 3S)-3-bromo-2-butanol
B) (2R, 3S)-2-bromo-3-butanol
C) (2R, 3S)-3-bromo-2-butanol
D) (2R, 3R)-2-bromo-3-butanol
E) (2R, 3R)-3-bromo-2-butanol
F) (2S, 3R)-2-bromo-3-butanol
G) (2S, 3R)-3-bromo-2-butanol
H) (2S, 3S)-2-bromo-3-butanol
CH3
HFOH
-Br
1) Provide the name for each structure shown.
NO ₂
2) Draw the structure of the molecule with the following name:
a) (1R, 3S)-Cyclopent-4-ene-1,3-diol
b) (2R, 3Z)-3-Chloro-2phenylhex-3-enal
c) (2E,4Z)-Hepta-2,4,6-trienoic acid
d) (3R, 4E)-3-isopropyl-6-oxohex-4-enenitrile
H₂C
NH
CH3
CH3
H₂N
CH3
The IUPAC name of the compound
is
(a) 5-formylhex-2-en-3-one
(b) 5-methyl-4-oxohex-2-en-5-al
(c) 3-keto-2-methylhex-5-enal
(d) 3-keto-2-methylhex-4-enal
The correct statement regarding electrophile is
(a) electrophile is a negatively charged species
and can form a bond by accepting a pair of
electrons from another electrophile
(b) electrophiles are generally neutral species
and can form a bond by accepting a pair of
electrons from a nucleophile
(c) electrophile can be either neutral or
positively charged species and can form a
bond by accepting a pair of electrons from
a nucleophile
(d) electrophile is a negatively charged species
and can form a bond by accepting a pair of
electrons from a nucleophile,
Which among the given molecules can exhibit
tautomerism?
Ph
Ph
I
III
(a) III only
(b) Both I and III
(d) Both II and III
(c) Both I and II
5)
Which of the following biphenyls is optically
active?
II
Chapter 7 Solutions
Organic Chemistry - Standalone book
Ch. 7.1 - Name each of the following using IUPAC...Ch. 7.1 - Prob. 2PCh. 7.2 - How many carbon atoms are sp2-hybridized in the...Ch. 7.3 - Prob. 4PCh. 7.3 - Are cis-2-hexene and trans-3-hexene stereoisomers?...Ch. 7.4 - Prob. 6PCh. 7.4 - Prob. 7PCh. 7.4 - Give the IUPAC name of each of the compounds in...Ch. 7.5 - Arrange the following in order of increasing...Ch. 7.6 - Prob. 10P
Ch. 7.6 - Standard enthalpies of formation are known for all...Ch. 7.6 - Prob. 12PCh. 7.6 - Despite numerous attempts, the alkene...Ch. 7.6 - Write structural formulas for the six isomeric...Ch. 7.7 - Place a double bond in the carbon skeleton shown...Ch. 7.9 - Identify the alkene obtained on dehydration of...Ch. 7.10 - Prob. 17PCh. 7.11 - Prob. 18PCh. 7.12 - Prob. 19PCh. 7.13 - The alkene mixture obtained on dehydration of...Ch. 7.14 - Write the structures of all the alkenes that can...Ch. 7.14 - Write structural formulas for all the alkenes that...Ch. 7.15 - A study of the hydrolysis behavior of...Ch. 7.15 - Use curved arrows to illustrate the electron flow...Ch. 7.15 - Predict the major product of the reaction shown.Ch. 7.16 - Prob. 26PCh. 7.17 - Prob. 27PCh. 7.18 - Prob. 28PCh. 7.19 - Predict the major organic product of each of the...Ch. 7.19 - A standard method for the synthesis of ethers is...Ch. 7 - Write structural formulas for each of the...Ch. 7 - Prob. 32PCh. 7 - Give an IUPAC name for each of the following...Ch. 7 - A hydrocarbon isolated from fish oil and from...Ch. 7 - Prob. 35PCh. 7 - Prob. 36PCh. 7 - Prob. 37PCh. 7 - Prob. 38PCh. 7 - Choose the more stable alkene in each of the...Ch. 7 - Suggest an explanation for the fact that...Ch. 7 - Prob. 41PCh. 7 - Write structural formulas for all the alkene...Ch. 7 - Prob. 43PCh. 7 - Prob. 44PCh. 7 - Predict the major organic product of each of the...Ch. 7 - Prob. 46PCh. 7 - Prob. 47PCh. 7 - The rate of the reaction In the first order in...Ch. 7 - Prob. 49PCh. 7 - Prob. 50PCh. 7 - You have available 2,2-dimethylcyclopentanol (A)...Ch. 7 - Prob. 52PCh. 7 - Prob. 53PCh. 7 - Prob. 54PCh. 7 - Acid-catalyzed dehydration of...Ch. 7 - The ratio of elimination to substitution is...Ch. 7 - Prob. 57PCh. 7 - Prob. 58DSPCh. 7 - Prob. 59DSPCh. 7 - Prob. 60DSPCh. 7 - Prob. 61DSPCh. 7 - A Mechanistic Preview of Addition Reactions The...Ch. 7 - Prob. 63DSPCh. 7 - Prob. 64DSPCh. 7 - Prob. 65DSP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The the following using IUPAC rules. Select the correct answer from the list below. Z O(Z)-3-methyl-4-propylhept-3-ene (E)-4-ethyl-3-methylhept-3-ene (E)-3-methyl-4-propylhept-3-ene (Z)-4-ethyl-3-methylhept-3-enearrow_forwardWhat is the proper IUPAC name for the compound whose structure is shown? Br A) (R)-3-bromohexane B) (S)-3-bromohexane C) (R)-4-bromohexane D) (S)-4-bromohexane E) None of thesearrow_forwardB The the following using IUPAC rules. Select the correct answer from the list below. Br (Z)-3-bromohex-2-ene (E)-4-bromohex-2-ene (E)-3-bromohex-2-ene O(Z)-4-bromohex-2-enearrow_forward
- The the following using IUPAC rules. Select the correct answer from the list below. Br (E)-4-bromohex-2-ene (Z)-4-bromohex-2-ene (Z)-3-bromohex-2-ene (E)-3-bromohex-2-enearrow_forwardH₂N- -Br H₂C Provide the locant and substituent in the order that they will be written in the name. The parent name is (old IUPAC version) or (new IUPAC version).arrow_forwardProvide the structures for each compound name given below, including notation for stereochemistry (i.e., R, S, E, Z, etc.) if necessary. a) N-ethylheptanamide b) methyl ethanoate c) 2,8-dimethyl-5-nonanonearrow_forward
- a) Draw the major product(s) of the following reactions. i) ii) OSO4 NaHSO4, H₂O BH3-THF H₂O₂, NaOH b) Methylcyclohexene reacts with chlorine both in CCl4 and water. i) Draw the structural formula for both the products. > ( )arrow_forwardThe IUPAC name of the compound H- is (a) 5-formylhex-2-en-3-one (b) 5-methyl-4-oxohex-2-en-5-al (c) 3-keto-2-methylhex-5-enal (d) 3-keto-2-methylhex-4-enal ( The correct statement regarding electrophile is (a) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile (b) electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile (c) electrophile can be either neutral or ar positively charged species and can form a bond by accepting a pair of electrons from a nucleophile (d) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from a nucleophile.. Which among the given molecules can exhibit tautomerism? Ph Ph I II III (a) III only (c) Both I and II (b) Both I and III (d) Both II and III 1) Which of the following biphenyls is optically active?arrow_forwardDraw the structure of the given compound. Identify if the given name is correct of incorrect. If not, provide the correct IUPAC name of the compound. Don’t use CIS/TRANS or E/Z in naming the compounds. 1) 6-PROPYLCYCOHEPTENE 2) 7-BUTYNYLDODEC-1-ENE Note: need answers immediately. Will give a good rate right away as well.arrow_forward
- Identify the correct IUPAC name for the following structure.A) 6-bromo-1-cyclopentyl-3,6-dimethylhexaneB) 1-bromo-5-cyclopentyl-1,4-dimethylpentaneC) 2-bromo-6-cyclopentyl-5-methylhexaneD) 5-bromo-1-cyclopentyl-2-methylhexaneE) none of the abovearrow_forward11) Name the IUPAC name of this compound? A) (R)(E)-2-phenyl-4-hexene B) (S)-(Z)-5-phenyl-2-hexene C) (R)-(Z)-2-phenyl-4-hexene D) (3)-(E)-5-phenyl-2-hexenearrow_forward-What of the following statements is false about cyclopentadienyl anion? A) It is aromatic B) It has 5 p molecular orbitals C) It has three full p bonding molecular orbitals D) It has one empty p anti-bonding molecular orbital E) It has two empty p anti-bonding molecular orbital 6. What is the correct IUPAC name of the following compound? A) 1-bromo-3-chloro-5-isobutyl-4-methylbenzene B) 5-bromo-1-chloro-3-sec-propyl-2-methylbenzene C) 5-bromo-1-sec-butyl-3-chloro-2-methylbenzene Br D) 1-bromo-3-sec-butyl-5-chloro-4-methylbenzene E) 5-bromo-1-chloro-3-isobutyl-2-methylbenzene II. Draw the structure for the major organic product of each of the following reaction: 7. CN + -> NC Show strerochemistry if necessary HBr 8. (1 equivalent) (45°C) Show strerochemistnuarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY