
Concept explainers
(a)
Interpretation:
The structure of chiral ether
Concept introduction:
A carbon atom that has four non-equivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The ethers contain
Answer to Problem 8.30AP
The structure of chiral ether
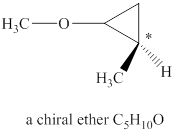
Explanation of Solution
The given molecular formula of ether is

Figure 1
The structure of chiral ether
(b)
Interpretation:
The structure of chiral alcohol with molecular formula
Concept introduction:
A carbon atom that has four non-equivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The alcohols contain hydroxyl
Answer to Problem 8.30AP
The structure of chiral alcohol with molecular formula
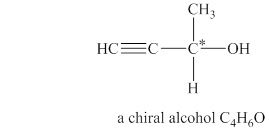
Explanation of Solution
The given molecular formula of alcohol is

Figure 2
The structure of chiral alcohol with molecular formula
(c)
Interpretation:
The structure of vicinal glycol
Concept introduction:
The alcohols contain hydroxyl
Answer to Problem 8.30AP
The structure of vicinal glycol
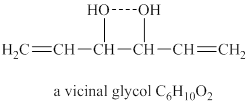
Explanation of Solution
The given molecular formula of vicinal glycol is

Figure 3
The structure of vicinal glycol
(d)
Interpretation:
The structure of diol
Concept introduction:
The alcohols contain hydroxyl
Answer to Problem 8.30AP
The structures of diol
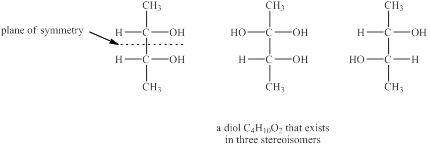
Explanation of Solution
The given molecular formula of diol is

Figure 4
The structures of diol
(e)
Interpretation:
The structure of diol
Concept introduction:
The alcohols contain hydroxyl
Answer to Problem 8.30AP
The structures of diol
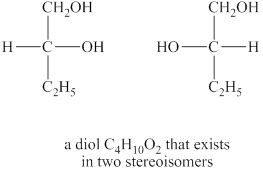
Explanation of Solution
The given molecular formula of diol is
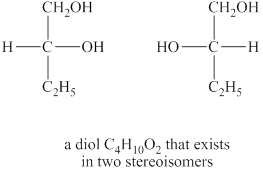
Figure 5
The structures of diol
(f)
Interpretation:
The structures of six
Concept introduction:
The ethers contain
Cyclic ethers with three-membered ring are commonly known as epoxide in which two carbons and one oxygen atom are arranged in cyclic form.
Answer to Problem 8.30AP
The structures of six epoxides (counting stereoisomers) with the molecular formula
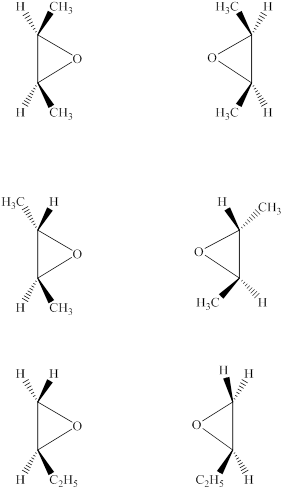
Explanation of Solution
The given molecular formula of the epoxide is

Figure 6
The structures of six epoxides (counting stereoisomers) with the molecular formula
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry
- (a) Draw the nine isomers having the formula C7H16 . (Hint: There is one structure with a seven-carbon chain, two structures with six-carbon chains [one is illustrated in the margin above], five structures with a five-carbon chain, and one structure with a fourcarbon chain.)(b) Identify the isomers of C7H16 that are chiral.arrow_forwardDraw the structural formulas for the following compounds. Include all the bonds to hydrogen atoms. Be sure to answer both parts. (a) 1,2,4-trimethylbenzene: (b) chlorobenzene:arrow_forwardDraw structural formulas for the following the compounds: (a) Cis-1,3-diphenylcyclohexane (b) 5-phenylpentanoic acid (c) 3,4-dibromo-N,N-dimethylanilinearrow_forward
- 2,3-Dibromoprop-1-ene (C3H4Br2) has four H atoms. Suppose that any of these H atoms can be replaced by a Cl atom to yield a molecule with the formula C3H3Br,Cl. (a) Identify two H atoms where this substitution would yield constitutional isomers of C3H3Br,CI; (b) enantiomers of C3H3Br,Cl; (c) diastereomers of C3H3Br,Cl. H нн H Br Br 2,3-Dibromoprop-1-enearrow_forwardDraw the structures for the following compounds.(a) 3-benzyl-4-bromohexane , 4,4-dimethylcyclohexene(b) trans-4,5-dibromohex-2-ene , cis-1,1-dibromo-2-ethyl-2,3-dimethylcyclobutanearrow_forwardDraw the structures of the following compounds:(a) Ethanoic acid(b) Bromopentane(c) Butanonearrow_forward
- Biphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.arrow_forward(d) This structure is that of an alcohol with the hydroxyl group on the third C atom of a five-carbon chain.The compound is called pentan-3-olarrow_forward(4) Write the structure of the following compounds from their IUPAC names. (a) ethanamide (b) methylethanoate (c) propanoic acid (d) 2-butanone (5) Write the structure of the following ethers and amines from their common names (a) methyl propyl (b) trimethylamine (6) Decide whether the following alcohols are polar or nonpolar (a) CH3CH2CH2CH2CH2CH2CH2OH (b) CH3CH2OHarrow_forward
- Write the bond line formula of the following compounds: (a) 4-methyl-2-hexene, two geometrical (stereoisomers) isomers (b) 3-fluoro-2-methylheptanol (3-fluoro-2-methylheptan-1-ol) (c) 4-methyl-hex-1-yn-3-olarrow_forwardDraw a structural formula of an alkene that undergoes acid-catalyzed hydration to give each alcohol as the major product (more than one alkene may give each alcohol as the major product). (a) 3-Hexanol (b) 1-Methylcyclobutanol (c) 2-Methyl-2-butanol (d) 2-Propanolarrow_forward(b) NABH, CH3 COCH,CH3 CH3CH2OH (c)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





