
Interpretation:
A balanced equation for the given hypothetical reaction is to be written, and the oxidation and hybridization state of sulfur in
required and the mass of
produced is to be calculated.
Concept introduction:
The oxidation state of an element is zero. The sum of the oxidation states of all the elements is equal to zero in a molecule of a compound, and in case of an ion, it is equal to the charge on the ion. Oxygen has a fixed oxidation state of
in its compounds.
To find hybridisation of an atom in a molecule, at first draw the Lewis structure of the molecule. Find the number of electrons domains around the atom. This gives the number of hybrid orbitals required for bonding. The number of hybrid orbitals is equal to the number of atomic orbitals that hybridise. Thus, one
and one
orbital hybridize to form two
and two
orbitals hybridize to form three
hybrid orbitals, and one
and three
orbitals hybridize to form four
hybrid orbitals.
The conversion factor is a fraction that is used to convert one unit to another. Use of more than one factor to find a solution is called dimensional analysis.
Answer to Problem 115AP
Solution: The balanced equation for the reaction is as follows:
The oxidation state of sulfur in elemental sulfur
is
is
is
is
is
and mass of
produced is
Explanation of Solution
The hypothetical reaction between elemental sulfur and sulfur trioxide is as follows:
The balanced equation for this reaction is as follows:
In
The oxidation state of oxygen is
Let
is the oxidation state of sulfur.
Then
is calculated as follows:
The oxidation state of sulfur in
is
In
Let
is the oxidation state of sulfur and the oxidation state of oxygen is
is calculated as follows:
The oxidation state of sulfur in
is
The Lewis structure of
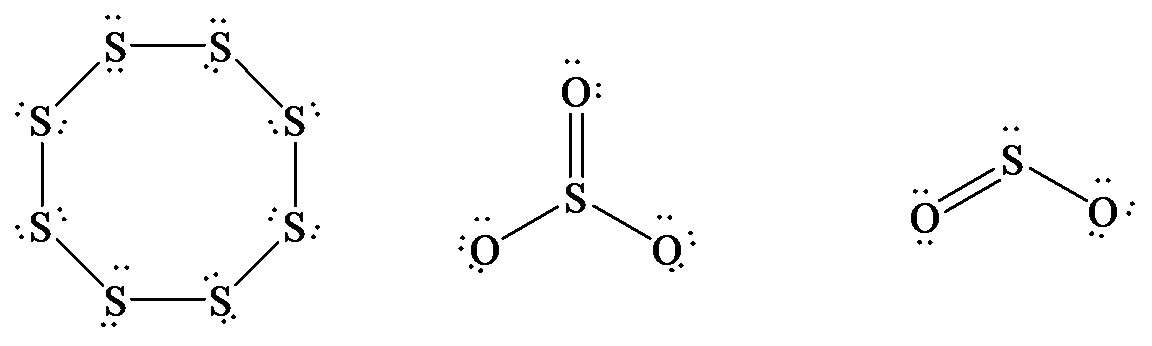
In
hybrid orbitals.
In
hybrid orbitals.
In
hybrid orbitals.
Consider the balanced equation:
One mole of
combines with
moles of
moles of
is
is given as
Convert the mass of
to moles as follows:
One mole of
combines with
moles of
Thus, for
needed is calculated as follows:
Molar mass of
is
Thus, the amount of
One mole of
produces
moles of
Thus, for
produced is calculated as follows:
Molar mass of
is
Thus, the amount of
Hence, mass of
required is
produced is
The balanced equation for the reaction between elemental sulfur and sulfur trioxide to produce sulfur dioxide is written; the oxidation and hybridization state of sulfur in
required and the mass of
produced by
of elemental sulfur are calculated.
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry
- Sketch the resonance structures for the N2O molecule. Is the hybridization of the N atoms the same or different in each structure? Describe the orbitals involved in bond formation by the central N atom.arrow_forwardThe three most stable oxides of carbon are carbon monoxide (CO), carbon dioxide (CO2), and carbon suboxide (C3O2). The space-filling models for these three compounds are For each oxide, draw the Lewis structure, predict the molecular structure, and describe the bonding (in terms of the hybrid orbitals for the carbon atoms).arrow_forwardSuppose you carry out the following reaction of ammonia and boron trifluoride in the laboratory. (a) What is the geometry of the boron atom in BF3? In H3NBF3? (b) What is the hybridization of the boron atom in the two compounds? (c) Considering the structures and bonding of NH3 and BF3, why do you expect the nitrogen on NH3 to donate an electron pair to the B atom of BF3? (d) BF3 also reacts readily with water. Based on the ammonia reaction above, speculate on how water can interact with BF3.arrow_forward
- For each of the following molecules or ions that contain sulfur, write the Lewis structure(s), predict the molecular structure (including bond angles), and give the expected hybrid orbitals for sulfur. a. SO2 b. SO3 c. d. e. SO32 f. SO42 g. SF2 h. SF4 i. SF6 j. F3SSF k. SF5+arrow_forwardIdentify the hybrid orbitals used by boron in BCl3 and in BCl4, the ion formed from the reaction of BCl3 and Cl. Explain your choices.arrow_forwardThe ionization energy of O2 is smaller than the ionization energy of atomic O; the opposite is true for the ionization energies of N2 and atomic N. Explain this behavior in terms of the molecular orbital energy diagrams of O2 and N2.arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





