
Concept explainers
Interpretation:
The volume of the atoms in bcc and fcc unit cell in comparison to the volume of the unit cell itself has to be calculated. From these data, the fraction of space occupied by atoms in bcc and fcc unit cell has to be calculated.
Concept Introduction:
Bcc unit cell:
Eight atoms occupy the corner position of a cube each contributing
Relationship between unit cell edge length and radius of a unit cell of a bcc arrangement:
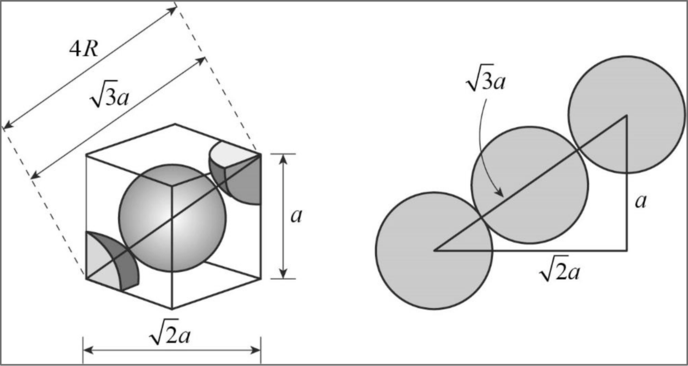
Figure 1
Applying Pythagoras theorem, from the diagram we can clearly concluded that
Body diagonal
Fcc unit cell:
Eight atoms occupy the corner position of a cube each contributing
Relationship between unit cell edge length and radius of a unit cell of a fcc arrangement:
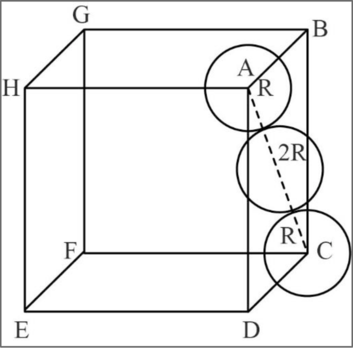
Figure 2
In fcc, the corner spheres are in touch with the face centered sphere as shown in the above figure. Hence, the face diagonal
Consider right angled triangle ACD.
Volume of a cubic unit cell:
Volume of cubic unit cell
Volume of a sphere:
Volume of a sphere
Packing fraction:
Packing fraction is the fraction of space occupied by total number of atoms per unit cell.
Mathematically, it can be represented as given below.
Fraction of space occupied by atoms in a unit cell
Where,
Answer to Problem 115QRT
The fraction of space occupied by atoms in bcc and fcc unit cell is
Explanation of Solution
The atoms can be assumed to be spherical in shape with radius
There are two atoms present per one bcc unit cell. The relationship between unit cell edge length and radius of a unit cell of a bcc arrangement is given below.
Now, the total volume of two atoms present in a bcc unit cell can be calculated as given below.
There are four atoms present per one fcc unit cell. The relationship between unit cell edge length and radius of a unit cell of a fcc arrangement is given below.
Now, the total volume of two atoms present in a bcc unit cell can be calculated as given below.
Volume of cubic unit cell ,
Now, the fraction of space occupied by atoms can be calculated as given below.
For bcc unit cell:
Fraction of space occupied by atoms
For fcc unit cell:
Fraction of space occupied by atoms
Therefore, the fraction of space occupied by atoms in bcc and fcc unit cell is
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry: The Molecular Science
- The CsCl structure is a simple cubic array of chloride ions with a cesium ion at the center of each cubic array (see Exercise 69). Given that the density of cesium chloride is 3.97 g/cm3, and assuming that the chloride and cesium ions touch along the body diagonal of the cubic unit cell, calculate the distance between the centers of adjacent Cs+ and Cl ions in the solid. Compare this value with the expected distance based on the sizes of the ions. The ionic radius of Cs+ is 169 pm, and the ionic radius of Cl is 181 pm.arrow_forwardCalculate the percent of volume that is actually occupied by spheres in a body-centered cubic lattice of identical spheres You can do this by first relating the radius of a sphere, r, to the length of an edge of a unit cell, l. (Note that the spheres do not touch along an edge but do touch along a diagonal passing through the body-centered sphere.) Then calculate the volume of a unit cell in terms of r. The volume occupied by spheres equals the number of spheres per unit cell times the volume of a sphere (4r3/3).arrow_forwardA portion of the crystalline lattice for potassium is illustrated below. (a) In what type of unit cell are the K atoms arranged? A portion of the solid-state structure of potassium. (b) If one edge of the potassium unit cell is 533 pm, what is the density of potassium?arrow_forward
- Crystalline polonium has a primitive cubic unit cell, lithium has a body-centered cubic unit cell, and calcium has a face-centered cubic unit cell. How many Po atoms belong to one unit cell? How many Li atoms belong to one unit cell? How many Ca atoms belong to one unit cell? Draw each unit cell. Indicate on your drawing what fraction of each atom lies within the unit cell.arrow_forwardCalculate the percent of volume that is actually occupied by spheres in a face-centered cubic lattice of identical spheres. You can do this by first relating the radius of a sphere, r, to the length of an edge of a unit cell, l. (Note that the spheres do not touch along an edge but do touch along the diagonal of a face.) Then calculate the volume of a unit cell in terms of r. The volume occupied by spheres equals the number of spheres per unit cell times the volume of a sphere (4r3/3).arrow_forwardConsider the three types of cubic units cells. (a) Assuming that the spherical atoms or ions in a primitive cubic unit cell just touch along the cubes edges, calculate the percentage of occupied space within the unit cell. (Recall that the volume of a sphere is (4/3)r3, where r is the radius of the sphere.) (b) Compare the percentage of occupied space in the primitive cell (pc) with the bcc and fcc unit cells. Based on this, will a metal in these three forms have the same or different densities? If different, in which is it most dense? In which is it least dense?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





