
(a)
Interpretation:
The effect of condensation temperature on the coefficient of performance is to be determined for different condensation temperatures assuming isentropic compression of vapor.
Concept introduction:
Below shown diagram represents vapor-compression refrigeration cycle on a
The line
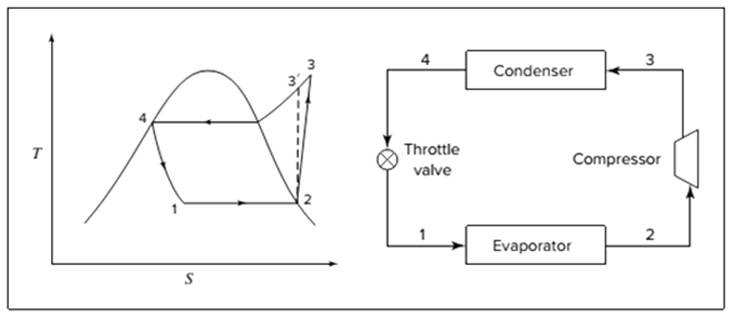
The equations used to calculate the heat absorbed in evaporator and the heat rejected in condenser are:
The work of compression is:
The coefficient of performance is:
The rate of circulation of refrigerant,
For Carnot refrigeration cycle, highest possible value of
(a)
Answer to Problem 9.13P
The coefficient of performance for condensation temperature of
The coefficient of performance for condensation temperature of
The coefficient of performance for condensation temperature of
As the condensation temperature is increased, the coefficient of performance decreases.
Explanation of Solution
Given information:
In a refrigerator, tetrafluoroethene acts as a refrigerant and operates with an evaporation temperature of
From table 9.1, the values of
Assume that the compressor efficiency is
For condensation temperature of
The saturation pressure at point 4 is the pressure at which the vapor condenses and
Also,
For isentropic compression,
At point
Using equation (4), the coefficient of performance for condensation temperature of
For condensation temperature of
The saturation pressure at point 4 is the pressure at which the vapor condenses and
Also,
For isentropic compression,
At point
Using equation (4), the coefficient of performance for condensation temperature of
For condensation temperature of
The saturation pressure at point 4 is the pressure at which the vapor condenses and
Also,
For isentropic compression,
At point
Using equation (4), the coefficient of performance for condensation temperature of
As the condensation temperature is increased, the coefficient of performance decreases.
(b)
Interpretation:
The effect of condensation temperature on the coefficient of performance is to be determined for different condensation temperatures assuming compressor efficiency of
Concept introduction:
Below shown diagram represents vapor-compression refrigeration cycle on a
The line
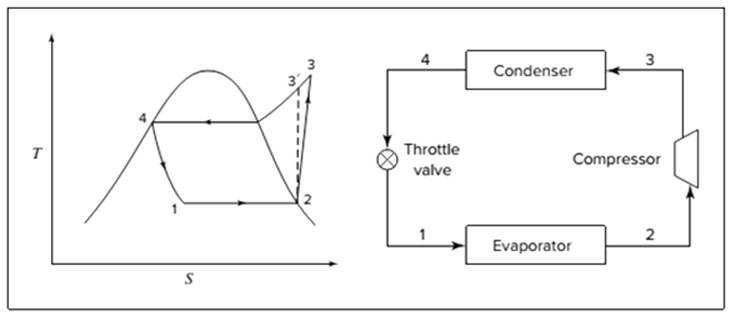
The equations used to calculate the heat absorbed in evaporator and the heat rejected in condenser are:
The work of compression is:
The coefficient of performance is:
The rate of circulation of refrigerant,
For Carnot refrigeration cycle, highest possible value of
(b)
Answer to Problem 9.13P
The coefficient of performance for condensation temperature of
The coefficient of performance for condensation temperature of
The coefficient of performance for condensation temperature of
As the condensation temperature is increased, the coefficient of performance decreases.
Explanation of Solution
Given information:
In a refrigerator, tetrafluoroethene acts as a refrigerant and operates with an evaporation temperature of
From table 9.1, the values of
The compressor efficiency is given as,
For condensation temperature of
The saturation pressure at point 4 is the pressure at which the vapor condenses and
Also,
For isentropic compression,
At point
Calculate
Now, calculate
Using equation (4), the coefficient of performance for condensation temperature of
For condensation temperature of
The saturation pressure at point 4 is the pressure at which the vapor condenses and
Also,
For isentropic compression,
At point
Calculate
Now, calculate
Using equation (4), the coefficient of performance for condensation temperature of
For condensation temperature of
The saturation pressure at point 4 is the pressure at which the vapor condenses and
Also,
For isentropic compression,
At point
Calculate
Now, calculate
Using equation (4), the coefficient of performance for condensation temperature of
As the condensation temperature is increased, the coefficient of performance decreases.
Want to see more full solutions like this?
Chapter 9 Solutions
Introduction to Chemical Engineering Thermodynamics
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





