
Concept explainers
Interpretation:
The atomic orbital overlap and MO energy diagrams for
Concept introduction:
According to
The molecular orbitals are formed by overlapping of atomic orbitals of adjacent atoms. The numbers of molecular orbitals formed are equal to the number of atomic orbitals overlap. The two atomic orbitals, on mixing along bonding axes, form two molecular orbitals, one is
Answer to Problem D.15YT
The atomic orbital overlap and MO energy diagrams for
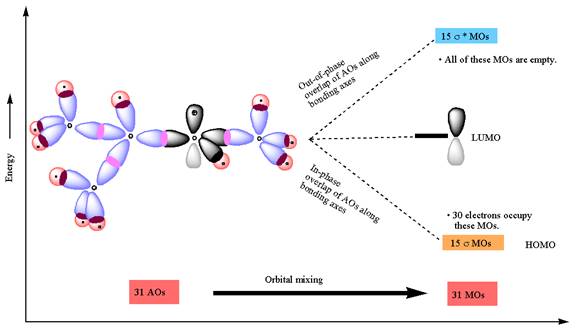
The constructed MO energy diagrams for
Explanation of Solution
In
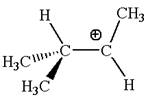
In forming the
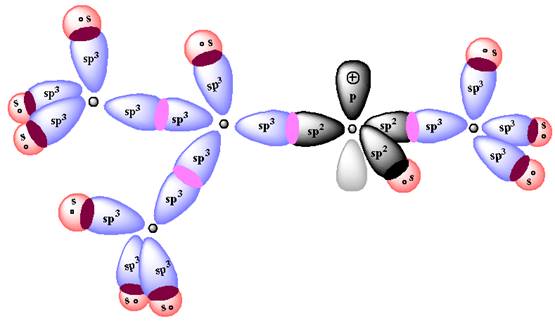
According to Aufbau Principle, the
The energy diagram for the formation of
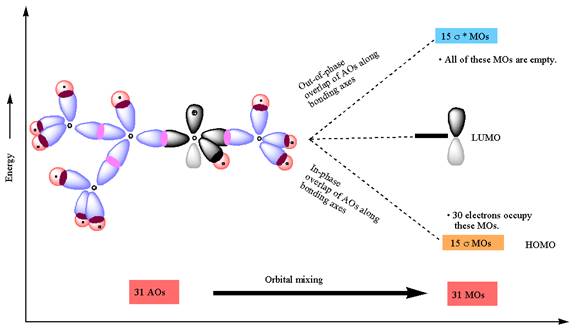
The Figure

The constructed MO energy diagrams for
The energy diagram for the formation of
Want to see more full solutions like this?
Chapter D Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- A reaction can take place and a new bond is formed when an electron rich species attacks an electron poor species. With this in mind, explain the following molecular orbital energy diagram. The "n" orbital refers to the nonbonding electrons on oxygen (electron rich), which is slightly higher in energy than the bonding electrons in the C=0 bond. Apply what you know, and think a little outside of the box.arrow_forwardA type of hyperconjugation occurs in the F-CH2-NH¯ anion even though there are no double bonds, triple bonds, or empty p AOs. Draw the orbital interaction that illustrates the most significant hyperconjugation interaction in this species. Hint: What is the HOMO in this species? Which adjacent o* orbital is the lowest in energy?arrow_forward7. Draw a picture showing the orbitals involved in the bonds only of the following. Hint: first draw the full Lewis Structure! (a) CH3CN (b) CH₂=CHCH₂CH=Oarrow_forward
- 661 10. Use the dash-wedge method to draw the 3-D structure of ammonia. a) What is the name of this type of structure? Trigonal Pyramidel b) What is the hybridization state of the N? S २० 11. Label all the nitrogens as 1º, 2º, or 3° then place the following labels on the molecules: (Aromatic or Nonaromatic) and (Amine or Amide). Aminc Aromatic 2⁰ 030 Amide nonaromatic N H-N-H Z-H More acidic hydrogens are present in pyrrole. 00 H-N-H CH3 10 Model 3: Amine Alkylation H N. CH₂CH3 CH₂2CH3 H-N-CH3 Br-CH3 BJ CH3 Br-CH3 Br-CH3 nonaromatic fast RXN 1A faster RXN 2A fastest Amine Nonoromatic RXN 3A NH₂ 12. Which of the following aromatic compounds (pyridine or pyrrole) is more basic? Explain. (Hint-think about the hybridization of each atom) N +! H-N-H CH3 -Z-H H (+)ī H-N-CH₂ CH3 2⁰ NH₂ 30 I I-Z: Aminc nonaromatic H IZ H N H-N-H 30 Amine Aromatic H-N-H CH3 0 H +i H3C-N-CH, H-N-CH3 CH3 CH3 O NH N. RXN 1B RXN 2B 4\\\\ RXN 3B २० ភ H-N-H 1 CH3 -H NH Amine Non-aromatic bb b po IZ Amine Nonaromatic…arrow_forwardHelp!! Phosgene, Cl2C=O, has a smaller dipole moment than formaldehyde, H2C=O, even though it contains electronegative chlorine atoms in place of hydrogen. Provide an explanation for this observation.arrow_forwardChapter 2 [References] H3C HB CH3 На, CH3 CH3 D H3C B НО Cholestanol differs from cholesterol only in the absence of a double bond in ring B. Draw the three-dimensional structure of cholestanol, and then determine the orientation of the following groups: #1: H at the junction of rings C & D with respect to ring C| #2: H at the junction of rings A & B with respect to ring B #3: Methyl at the junction of rings C & D with respect to ring C| O Iarrow_forward
- Place the following in order of decreasing dipole moment. |- CH4 II - CH3OH III - CH3SH III > | > || Il > | > II | > II| > | || > III > I | = II| > ||arrow_forwardThe partial Lewis structure below is an example of an amide functional group, which we will learn about in the next chapter. What is the hybridization and geometry of the methyl (CH3) carbon? What is the hybridization and geometry of the central carbon? (Note: the structure below is intended to show connectivity only - the geometry as drawn is not representative of the true structure!) CH3–C-NH, Next Previousarrow_forwardIn the sketch of the structure of SO2 label all bonds. Drag the appropriate labels to their respective targets. Labels can be used once, more than once, or not at all. : S(p) – O(p) Lone pair in p orbital Lone pair in sp? orbital o : S(p) – 0(sp²) т: S(p) — О(p) T: S(sp²) – O(p) r: S(sp²) – O(p) S(p) – O(sp²)arrow_forward
- A student argues that the two nitrogens in the compound below are sp? hybridized, but atom a is still more basic than atom b. Is the student correct? Explain. •• -N-CH2CH3 | b CH2CH3 aarrow_forward8. Assign Z and E configurations to these compounds. O HO- HO O CIarrow_forwardThe most stable MO of 1,3,5-hexatriene and the most stable MO of benzene are shown here. Which compound is more stable? Why?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
