
Introduction to General, Organic and Biochemistry
11th Edition
ISBN: 9781285869759
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 12, Problem 12.62P
Interpretation Introduction
Interpretation:
The possible number of geometrical (cis/trans) isomers in linolenic acid (a fatty acid) should be determined.
Concept Introduction:
The structure of linolenic acid is.
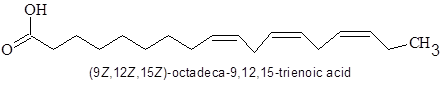
So there are three cis-double bonds.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What kind of fatty acid is the following structure?
||
HO-C-(CH2)14—CH3
monounsaturated
Opolyunsaturated
saturated
Is a fatty acid with the following
structure saturated or unsaturated?
CH3-CH,-CH, C-OH
|3D
Ω
Which of the following is TRUE about amylopectin?
- it is a linear polysaccharide composed of D-glucose
- accounts for about 20% by weight of starch
accounts for about 80% by weight of cellulose
- accounts for about 80% by weight of starch
- it is a branched polysaccharide of galactose
Refer to the molecular formula for the following fatty acids:
A. CH3(CH2)10CO2H
B. CH3(CH2)12CO2H
C. CH3(CH2)14CO2H
D. CH3(CH2)16CO2H
Е. СН3(СH2)18СО2Н
Which of the following statement is TRUE?
- Fatty acid C could have the lowest melting point.
Fatty acid A could have the highest melting point.
- Fatty acid E could have the highest melting point.
- Fatty acid D could have the lowest melting point.
Refer to the structures of carboxylic acid and derivatives. If pure compound B reacts with pure
water, which of the following statements is TRUE?
H,N
CH3 H,C
CH, H,C"
CH3
CH
A.
B
D
E
The product of the reaction is compound A.
The product of the reaction is compound C.
The product of the reaction is compound D…
Chapter 12 Solutions
Introduction to General, Organic and Biochemistry
Ch. 12.3 - Prob. 12.1PCh. 12.3 - Prob. 12.2PCh. 12.3 - Problem 12-3 Write the IUPAC name for each...Ch. 12.3 - Problem 12-4 Draw structural formulas for the...Ch. 12.3 - Problem 12-5 How many stereoisomers are possible...Ch. 12.5 - Prob. 12.6PCh. 12.5 - Problem 12-7 Propose a two-step mechanism for the...Ch. 12.5 - Prob. 12.8PCh. 12.5 - Problem 12-9 Propose a three-step reaction...Ch. 12.5 - Prob. 12.10P
Ch. 12 - Prob. 12.11PCh. 12 - Answer true or false. Both ethylene and acetylene...Ch. 12 - 12-13 What is the difference in structure between...Ch. 12 - There are three compounds with the molecular...Ch. 12 - 12-15 Name and draw structural formulas for all...Ch. 12 - Prob. 12.16PCh. 12 - Draw a structural formula for at least one...Ch. 12 - Each carbon atom in ethane and in ethylene is...Ch. 12 - Prob. 12.19PCh. 12 - Prob. 12.20PCh. 12 - Prob. 12.21PCh. 12 - 12*22 Draw a structural formula for each compound....Ch. 12 - 12-23 Draw a structural formula for each compound....Ch. 12 - Prob. 12.24PCh. 12 - 12-25 Write the IUPAC name for each unsaturated...Ch. 12 - Explain why each name is incorrect and then write...Ch. 12 - 12-27 Explain why each name is incorrect and then...Ch. 12 - Prob. 12.28PCh. 12 - 12-29 Which of these alkenes show cis-trans...Ch. 12 - 12-30 Which of these alkenes shows cis-trans...Ch. 12 - 12-31 Cyclodecene exists as both cis and trans...Ch. 12 - Arachidonic acid is a naturally occurring C„o...Ch. 12 - Prob. 12.33PCh. 12 - If you examine the structural formulas for the...Ch. 12 - 12*35 For each molecule that shows eis-trans...Ch. 12 - Name and draw structural formulas for all...Ch. 12 - /3-Ocimene, a triene found in the fragrance of...Ch. 12 - Answer true or false. Alkenes and alkynes are...Ch. 12 - Prob. 12.39PCh. 12 - 12-40 Define alkene addition reaction. Write an...Ch. 12 - Prob. 12.41PCh. 12 - 12-42 Complete these equations.Ch. 12 - Draw structural formulas for all possible...Ch. 12 - Prob. 12.44PCh. 12 - 12-45 Draw a structural formula for the product of...Ch. 12 - Draw a structural formula for an alkene with the...Ch. 12 - 12-47 Draw a structural formula for an alkene with...Ch. 12 - Draw a structural formula for an alkene with the...Ch. 12 - Prob. 12.49PCh. 12 - 12-50 Draw the structural formula of an alkene...Ch. 12 - Prob. 12.51PCh. 12 - Prob. 12.52PCh. 12 - Following is the structural formula of...Ch. 12 - Propose an explanation for the following...Ch. 12 - There are nine alkenes with the molecular formula...Ch. 12 - Prob. 12.56PCh. 12 - 12-57 Hydrocarbon A, Cf,Hs, reacts with 2 moles of...Ch. 12 - 12-58 Show how to convert ethylene to these...Ch. 12 - 12-59 Show how to convert 1-butene to these...Ch. 12 - Prob. 12.60PCh. 12 - 12-61 (Chemical Connections 12A) What is one...Ch. 12 - Prob. 12.62PCh. 12 - Prob. 12.63PCh. 12 - 12-64 (Chemical Connections 120 What is the...Ch. 12 - (Chemical Connections 120 Assume that 1 X IO-12 g...Ch. 12 - Prob. 12.66PCh. 12 - 12-67 (Chemical Connections 12D ) In which isomer...Ch. 12 - Prob. 12.68PCh. 12 - Prob. 12.69PCh. 12 - Prob. 12.70PCh. 12 - Prob. 12.71PCh. 12 - Prob. 12.72PCh. 12 - Prob. 12.73PCh. 12 - Propose a structural formula for the product!s)...Ch. 12 - Prob. 12.75PCh. 12 - Draw the structural formula of an alkene that...Ch. 12 - 12-77 Show how to convert cyclopentene into these...Ch. 12 - Prob. 12.78PCh. 12 - Prob. 12.79PCh. 12 - In omega-3 fatty adds, the last carbon of the last...Ch. 12 - Prob. 12.81P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The structural formula for the open-chain form of galactose is CH Н—ҫ—ОН Но—С—н НО—С—Н Н—ҫ—ОН CH-ОН (a) Is this molecule a sugar? (b) How many chiral carbons are present in the molecule? (C) Draw the structure of the six-member-ring form of this molecule.arrow_forwardThis molecule (is or is not) a reducing sugar. The sugar molecule is attached to the alcohol at carbon (just use a number) in the steroid structure. The lactone group is attached to carbon (just use a number) in the steroid structure. There are (just use a number) chiral carbons in this molecule. CH3 HỌ но. но, H A ÕH но ÕH CH3 • 8H2O ОН ОНarrow_forward1. Abundant trans bonds make partially hydrogenated vegetable oil a very unhealthy food choice. Vegetable oil can also be hydrogenated until it becomes fully saturated with hydrogen atoms. Would the physical properties of the hydrogenated and partially hydrogenated oils differ? If so, how and why would the differences occur? Do you think that full hydrogenation makes vegetable oil more or less healthy to eat, or does it have no effect? 2. Lipoprotein particles are relatively large, spherical clumps of protein and lipid molecules (see Figure 3.18) that circulate in the blood of mammals. They are like suitcases that move cholesterol, fatty acid remnants, triglycerides, and phospholipids from one place to another in the body. Given what you know about the insolubility of lipids in water, which of the four kinds of lipids would you predict to be on the outside of a lipoprotein clump, bathed in the water-based fluid portion of blood? 3. In 1976, a team of chemists in the United Kingdom was…arrow_forward
- a In omega-3 fatty acids, the last carbon of the last double bond of the hydrocarbon chain ends three carbons from the methyl terminal end of the chain. The last carbon of the chain is called the omega carbon, hence, the designation omega-3. Eicosapentaenoic acid is a common omega-3 fatty acid found in cold water fatty fish and health food supplements. How many cis-trans isomers are possible for this fatty acid? cis-trans isomersarrow_forwardWhich of the following structures is a 20:2 (Δ11,16) fatty acid? a. CH3(CH2)9CH=CH(CH2)3CH=CH(CH2)2COOHb. CH3(CH2)2CH=CH(CH2)3CH=CH(CH2)9COOHc. CH3(CH2)10CH=CH(CH2)3CH=CHCH2COOHd. CH3CH2CH=CH(CH2)3CH=CH(CH2)10COOHarrow_forward1. Some marine plankton contain triacylglycerols formed from the polyunsaturated fatty acid (PUFA) such as: CH3CH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2COOH a) Draw the skeletal structure of the given PUFA showing the preffered stereogenic arrangement for a naturally-occuring lipid at each double bond. b) Write the systematic name of the fatty acid. c) What type of omega-n acid is this fatty acid? d) Would you expect this fatty acid to be a soild or liquid at room temperature?arrow_forward
- Consider the following fatty acids: CH3-(CH₂) 12-C-OH Myristic Acid CH3-(CH2)4-CH=CH-CH₂-CH=CH-CH2- 0 B) Which fatty acid listed above would have the lower melting point? Linoleic Acid A) What type of fatty acid is Linoleic Acid (Saturated, Monounsaturated or Polyunsaturated)? C) Draw the structure of a triacylglycerol that contains two molecules of linoleic acid and one molecule of myristic acid D) True or False: As the number of carbons in a saturated fatty acid increases, the melting point decreases. E) True or False: As the number of double bonds in an unsaturated fatty increases the melting point decreases. A/Carrow_forwardIndicate whether each statement is true or false: (a) Trans fats are saturated. (b) Fatty acids are long-chain carboxylic acids. (c) Monounsaturated fatty acids have one CC single bond in the chain, while the rest are double or triple bonds.arrow_forwardXylulose has the following structural formula. To what carbohydrate class does xylulose belong based on the number of carbons and carbonyl functionality? A) aldotetrose B) aldopentose C) ketotetrose D) ketopentose E) ketohexosearrow_forward
- The structural formula for the open-chain form of D-mannose is CH НО—С—Н Но-с—н Н—С—ОН Н—ҫ—ОН CH-OН (a) Is this molecule a sugar? (b) How many chiral carbons are present in the molecule? (c) Draw the structure of the six-member-ring form of this molecule.arrow_forwardIf an unsaturated fatty acid has 18 carbons, a double bond at position ∆15 would be in ω notation ω - _________? The functional groups which hold fatty acid molecules and glycerol molecules together in triacylglecerol molecules are called: ____________ If a triacylglycerol is heated in aqueous sodium hydroxide (lye) solution glycerol and the sodium salts of fatty acids are produced. This mixture of sodium salts of fatty acids is called in common terminology ________. Name the four parts of phosphatidylcholine. _______, _________, ________ and _________ The molecule found in phospholipids which most resembles glucose is: a) lecithin b) inositol c) choline d) kinetic acid A molecule which contains sphingosine bonded to a fatty acid and a monosaccharide is called ________ A lipid class which contains no fatty acids is called ___________ When phosphatidylcholine (lecithin) is sonicated in water it forms ____________ Membrane fluidity may be regulated by the degree of…arrow_forwardHO-CH–CH=CH-(CH2)12-CH3 CH2 CH2 CH2 CH2 CH2 CH2 CH2 CH-NH-Ĉ–CH2 `CH2 `CH2 °CH2 `CH2 `CH2 `CH2 `CH2 || CH2-OH The structure above is consistent with what class of lipids? O Glycolipids Glycerophospholipids Sphingolipids none of the above O O O Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY