
a)
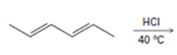
Interpretation:
The major products formed during the addition of one equivalent of HCl to hexa-2, 4-diene along with the mechanism of their formation is to be shown.
Concept introduction:
Conjugated dienes undergo electrophilic addition reactions through the formation of an allyl carbocation. The allyl cation is resonance stabilized and the attack of chloride ion on each of these forms leads to the formation of a mixture of 1, 2- and 1, 4-addition products.
To show:
The major product formed during the addition of one equivalent of HCl to hexa-2, 4-diene along with the mechanism of their formation.
b)
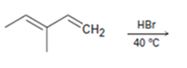
Interpretation:
The major products formed during the addition of one equivalent of HX to 3-methylpenta-1, 3-diene along with the mechanism of their formation is to be shown.
Concept introduction:
Conjugated dienes undergo electrophilic addition reactions through the formation of an allyl carbocation. The allyl cation is resonance stabilized and the attack of chloride ion on each of these forms leads to the formation of a mixture of 1, 2- and 1, 4-addition products.
To show:
The major products formed during the addition of one equivalent of HBr to 3-methylpenta-1, 3-diene along with the mechanism of their formation.
c)
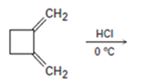
Interpretation:
The major product formed during the addition of one equivalent of HCl to 1, 2-dimethylenecyclobutane, along with the mechanism of their formation is to be shown.
Concept introduction:
Conjugated dienes undergo electrophilic addition reactions through the formation of an allyl carbocation. The allyl cation is resonance stabilized and the attack of chloride ion on each of these forms leads to the formation of a mixture of 1, 2- and 1, 4-addition products.
To show:
The major product formed during the addition of one equivalent of HCl to 1, 2-dimethylenecyclobutane along with the mechanism of their formation.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
- Provide the major product(s) for each reaction below and you may assume there is anexcess of each reagent for each reactionarrow_forwardProvide detailed step-wise mechanism for the following reaction. Be sure to show all intermediates, formal charges, and show the movement of electrons with curved arrows. a 4+H₂Q 044 OHarrow_forwardGive the major product(s) of the following reaction. NH3 HO- heat Harrow_forward
- 2- Mention the product of a nucleophilic substitution reaction of (S)-2-bromohexane with acetate ion, CH3CO2- Assume that inversion of configuration occurs, and show the chemistry of both the reactant and product. 3- Draw the mechanism of the SN2 Reaction. Show the correct directions of the arrows, and all reagents. 4- Draw the mechanism of the SN1 Reaction. Show the correct directions of the arrows, and all reagents. 5- Draw the mechanism of the E2 Reaction with an Alkyl Halide. Show the correct directions of the arrows, and all reagents. 6- Draw the mechanism of the E1 Reaction with an Alkyl Halide. Show the correct directions of the arrows, and all reagents. 7- Mention the product formed in an SN2 reaction between 1-bromobutane and NaI. 8- Rank the following compounds in order of their expected reactivity toward SN2 reaction: CH3Br, CH3OTos, (CH3)2CHCl. 9- Explain Grignard Reagents in details with one example. 10- Mention how a halogen substituent can be replaced by a deuterium atom…arrow_forward3 Give the major product of the following reaction. U O H OH CrO3 H₂SO4 ? OH O There is no reaction under these conditions or the correct product is not listed here.arrow_forwardGive the major product of the following reaction. HO 1) 03 CH3 2) H₂O OH There is no reaction under these conditions or the correct product is not listed here. CH₂ OH H ?arrow_forward
- Provide the major product for the reaction of the following starting material with excess HBr. If more than one product is formed, list the major product first. If no reaction, draw the starting material.arrow_forward1. Give the major product(s) for the following reactions. b) c) ОН I B. NO OCH 3 OH HNO3 H₂SO4 HNO3 H₂SO4 HNO3 H₂SO4arrow_forwardVI. Propose a mechanism for the following reaction. (rearrangement after initial protonation). CH=CH₂ CH=CH₂ H-Br HBF Br CH₂CH₂ iCH-CH3 снаснз Br Br ex میری sos a) Is the initially formed carbocation Primary, secondary or tertiary? b) Is the rearranged carbocation Primary, secondary or tertiary?arrow_forward
- Give the major product(s) of the following reaction. Cro3 H2SO4 H. HO. OH There is no reaction under these conditions or the correct product is not listed here.arrow_forwardGive the major product of the following reaction. 1) Hg(OAc)2, CH3OH ZON 2) NABH4, NAOH Br NO OMe Br NO, HO. NO2 Br NO, OMe There is no reaction under these conditions or the c ON Br NO2 OMe NO2arrow_forwardIdentify the major product of the following reaction sequence. 1) [H*], H₂NNH2, (-H₂O) 2) KOH/H₂O, heat HO₂C HO₂C HO CO,H OH OH There is no reaction under these conditions or the correct product is not listed here.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
