
Concept explainers
a)
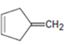
Interpretation:
Whether 4-methylenecyclopent-1-ene will have a π – π * UV adsorption in the 200 to 400nm range is to be stated.
Concept introduction:
Compounds having alternating double bonds (conjugated dienes) will have π – π *UV adsorption in the 200 to 400nm range.
To state:
Whether 4-methylenecyclopent-1-ene will have a π – π * UV adsorption in the 200 to 400nm range.
b)
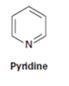
Interpretation:
Whether pyridine will have a π – π * UV adsorption in the 200 to 400nm range
Concept introduction:
Compounds having alternating double bonds (conjugated dienes) will have π – π * UV adsorption in the 200 to 400nm range.
To state:
Whether pyridine will have a π – π * UV adsorption in the 200 to 400nm range
c)
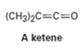
Interpretation:
Whether the ketene given will have a π – π * UV adsorption in the 200 to 400 nm range
Concept introduction:
Compounds having alternating double bonds (conjugated dienes) will have π – π * UV adsorption in the 200 to 400nm range.
To state:
Whether the ketene given will have a π – π * UV adsorption in the 200 to 400 nm range
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Organic Chemistry
- For the substituted cyclohexane compound shown, identify the atoms that are trans to the bromo substituent. I CH3 Br HD HCI- HA HB НОЕ K ČI HG Identify the atoms that are trans to the bromine. A В C D E F G H Karrow_forwardThe IR, and 1H NMR spectra of a compound with formula C5H6O2 are shown below. Determine the structure of the compound. You must fill all the parts to this question in the given spaces.arrow_forward1) A compound with molecular formula C49H96 could not be a ... 3)Compound Y has molecular formula C4H8O and its IR spectrum shows no absorption above 3000 cm-1 and no absorption between 1680 and 1840cm-1. Which of the following could be compound Y? 4) Which of the following will have its carbonyl absorption in its IR spectrum at the lowest wavelength? Please explain 1,3,4 thank youarrow_forward
- An unknown compound (whose molecular formula is C5H8O) shows IR absorption at 3600 and 3300 cm^-1. It’s ^1H-NMR spectrum contained singlets at delta 1.5, 2.2, and 2.9 (broad) ppm, in the ratio 6:1:1. Draw the structure for the unknown compound.arrow_forwardDraw the bond-line (skeletal) structure of the compound with molecular formula C₂H₁3Br that gives the following alkene as the exclusive product of E2 elimination. Click and drag to start drawing a structure. C X C Ś c+arrow_forwardWe have no way of predicting whether the reagent shown will add to the Re or Si face of the aldehyde functional group. Which of the following is true about the reaction shown? CH, CH3 LIC CH OH CH The reaction can produce a mixture of R,R; S,S; R,S; and S,R isomers. The reaction may yield a mixture of R,S and R,R diastereomers. The reaction may create a mixture of S,R and S,S diastereomers. O The reaction will yield either R,R or S,S enantiomers.arrow_forward
- 4. Which of the following compounds will not show optical activity? a) CH2CICHBrCI b) CHзCH(ОH)СНз с) CH-CH-CHCICНЗ d) CH:(ОH)CH(NH-)COОHarrow_forward19. Specify whether you expect the benzene rings in the following compounds to be activated or deactivated. NH₂ NO₂ b) F NO₂ OH NO₂ NO₂arrow_forwardA compound has the molecular formula C6H12O2. Its IR spectrum shows a strong absorption band near 1740 cm-1. Its 1H NMR spectrum consists of two singlets at δ 1.2 and δ 3.6. Which is the most likely structure of the compound?arrow_forward
- For the substituted cyclohexane compound shown, identify the atoms that are trans to the bromo substituent. I CH3 Br J HD HCI НА FF HB НОЕ HG CI Identify the atoms that are trans to the bromine. A В D E F H Karrow_forwardCH₂0- -CH, CH, CHỊCHO- 40-CHECH CH₂CH₂CH, CH₂0- 30- A B CH3 O None, they would all have identical UV-VIS spectra. OB OA C с Which compound(s), if any, can be distinguished from the others by their UV-VIS spectra? -CH₂CHarrow_forward● ● ● ● (A) Cl₂ CH₂=CHCH3 heat Propene OH HOCH₂CHCH₂OH 1,2,3-Propanetriol (glycerol, glycerin) Provide structures for all intermediate compounds (A-D) and describe the type of mechanism by which each is formed. (B) 2A (C₂H₂C1) CI a C (C₂H₂C1O₂) *85 H H₂C=C All hydrogen atoms are implied. Apply formal charges where appropriate. Omit lone pairs and radical electrons from your answer. You do not have to consider stereochemistry. C-Cl H₂ NaOH, H₂O internal displacement Lindlar reduction SN1-substitution SN2-substitution acid-catalyzed hydrolysis addition-elimination allylic halogenation halohydrination vinylic halogenation H H₂C=C Ca(OH)2 D (C3H6O₂) heat → B (C₂H₂O) 00 OU Cl₂, H₂O در H₂O, HCI 2 ster n ? ChemDoodle [Refere ?arrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

