
Chemistry In Focus
6th Edition
ISBN: 9781305084476
Author: Tro, Nivaldo J., Neu, Don.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 15, Problem 3SC
Interpretation Introduction
Interpretation:
The structures of ethyl-2-methylbutanoate and civetone are to be examined and upon opening a bottle containing the mixture of these two, which one would dominate initial smell is to be determined.
Concept introduction:
Civetone is an unsaturated macrocyclic
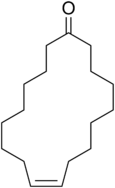
Civetone
Graham’s law states that the rate of diffusion is inversely proportional to the square root of mass of its particles.
Molar mass is defined as the ratio of moles
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Aldehydes have which type of smell?
Fish like smell
Pungent smell
Bitter almond smell
Rotten egg like smell
Organic chemistry:How do you account for the smell of vinegar when an old bottle of aspirin is opened ?
When diethyl ether is spilled on the skin, the skin takes on a dry appearance. Explain whether this effect is more likely due to a removal of water or a removal of natural skin oils
Chapter 15 Solutions
Chemistry In Focus
Ch. 15 - Prob. 15.1YTCh. 15 - Prob. 1SCCh. 15 - Prob. 2SCCh. 15 - Prob. 3SCCh. 15 - Prob. 1ECh. 15 - Prob. 2ECh. 15 - Prob. 3ECh. 15 - Prob. 4ECh. 15 - Prob. 5ECh. 15 - Prob. 6E
Ch. 15 - Prob. 7ECh. 15 - Prob. 8ECh. 15 - Prob. 9ECh. 15 - Prob. 10ECh. 15 - Prob. 11ECh. 15 - Prob. 12ECh. 15 - Prob. 13ECh. 15 - Define eutrophication.Ch. 15 - Prob. 15ECh. 15 - Prob. 16ECh. 15 - Prob. 17ECh. 15 - Prob. 18ECh. 15 - Prob. 19ECh. 15 - What are the three types of interactions that...Ch. 15 - Prob. 21ECh. 15 - Prob. 22ECh. 15 - Prob. 23ECh. 15 - Prob. 24ECh. 15 - Prob. 25ECh. 15 - Prob. 26ECh. 15 - Prob. 27ECh. 15 - Prob. 28ECh. 15 - Prob. 29ECh. 15 - How do sunscreens protect your skin from the Suns...Ch. 15 - Prob. 31ECh. 15 - Prob. 32ECh. 15 - Prob. 33ECh. 15 - Prob. 34ECh. 15 - Prob. 35ECh. 15 - Prob. 36ECh. 15 - Prob. 37ECh. 15 - Prob. 38ECh. 15 - Prob. 39ECh. 15 - Prob. 40ECh. 15 - Prob. 41ECh. 15 - Prob. 42ECh. 15 - Prob. 43ECh. 15 - Prob. 44ECh. 15 - Prob. 45ECh. 15 - Prob. 46ECh. 15 - The salt bridges that hold hair protein (keratin)...Ch. 15 - Prob. 48ECh. 15 - The hydrochloric acid present in toilet bowl...Ch. 15 - Prob. 50ECh. 15 - Prob. 51ECh. 15 - Prob. 52ECh. 15 - Prob. 53ECh. 15 - Prob. 54ECh. 15 - Prob. 55ECh. 15 - Prob. 56ECh. 15 - Prob. 57ECh. 15 - Prob. 58ECh. 15 - Prob. 59ECh. 15 - Prob. 60ECh. 15 - Prob. 61ECh. 15 - Prob. 62ECh. 15 - Prob. 64ECh. 15 - Prob. 65E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Ester compounds often have a sweet, pleasant odor. Many characteristic fruit scents are largely due to the natural presence of one or more ester compounds. As such, artificial scents for foods are often composed of complex mixtures of various esters. The exact identity and ratio of ingredients that compose a particular scent are closely guarded secrets in the food and fragrance industry. Suppose that you are a chemist working for a company that i creating a new line of air fresheners. The company is considering three scents: apple, pear, and pineapple. The project manager has asked you to prepare the ester compounds that are largely responsible for these scents. The structural formulas for these ester compounds are shown here: Alcohols for Air Freshener Project Molar mass Density Cost, per (g/mL) Reagent (g/mol) 1.00 L methanol 32.04 0.79 $46.20 ethanol 46.07 0.79 $112.00 1-propanol 60.10 0.80 $72.70 1-butanol 74.12 0.81 $72.60 Use the structural formulas of the alcohols and…arrow_forwardExplain why NOT ALL hydrolysis of esters produce a sour smellarrow_forwardUsing the relationship between the structure and the boiling point. Obtain a table for the listed chemicals below showing how the boiling point will be different with (1) the different chemical structures, (2) with different functional groups. Boiling Point and Functionalized Compounds: Chemical: Cyclohexanol Cyclohexanone Cyclohexene Benzaldehyde Benzyl alcoholarrow_forward
- What chemistry properties or structure make Salicylic acid cannot be taken orally? Is it due to the phenol functional group. Is hydroxyl (—OH) group attached to a carbon atom in a benzene ring what makes salicylic acid to be irritating? (please type clearly,please no blurry handwriting)arrow_forwardIf the fragrance-containing balloon were heated in a microwave oven for a few moments, would the smell be more or less pronounced? Why?arrow_forwardThese are a class of organic compounds that are easily vaporized and can remain suspended in the air as potent contaminants. sulfur oxides volatile organic compounds nitrogen oxides ozone depleting substances dioxins and furansarrow_forward
- Draw a schematic diagram to differentiate cyclohexanone, acetone and benzaldehyde using the five qualitative tests: 2,4-dinitrophenyl hydrazine test, Benedict’s test, Tollen’s test, Jones’ test, and Iodoform test.arrow_forwardDraw the skeletal ("line") structure of the smallest organic molecule that produces potassium 2-methylpropanoate when reacted with KOH. Click and drag to start drawing a structure. ☐:arrow_forwardWrite the chemical equation for a pentanoate ion acting as base when it reacts with hydrochloric acid (HCI). Which is formula of this rule of reaction?arrow_forward
- Two major components of a skunk’s scent fluid are 3-methyl-1-butanethiol and trans-2-butene-1-thiol. Write structural formulas for each of these compounds.arrow_forwardWhat is an amide? What is its general formula? How do you classify an amide? Explain What makes amide prevalent in nature? Cite some amides found in our body. What are the properties of amide? What are the factors that affect these properties? Draw 5 amide structures and write their IUPAC name.arrow_forwardWrite the chemical equation between acetic acid and sodium hydroxide.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning