
Concept explainers
Interpretation:
The cycloaddition reaction involving two molecules of cyclopentene is as follows:
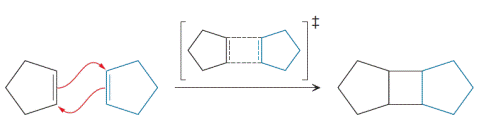
Whether the transition state is
Concept introduction:
The Diels-Alder reaction is a single step concerted reaction, whereby all of the bonds are formed and broken simultaneously. Thus, the reaction proceeds through a cyclic transition state, which makes it a pericyclic reaction. All Diels-Alder reactions involve the cyclic flow of six π electrons. The number of electrons is important because these electrons are delocalized over a complete ring in the transition state. Six electrons is a Hückel number of electrons (i.e., an odd number of pairs), and the species is aromatic when the electrons are delocalized over an entire ring. Thus, Diels-Alder reaction proceeds through an aromatic transition state.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- The conversion of reactant R to product A is under control and is preferred a temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, high The conversion of reactant R to product B is under control and is preferred at temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, high Electrophilicity is the tendency of a species to accept electrons due to energy orbitals. low, unoccupied low, occupied high, unoccupied high, occupiedarrow_forwardThe conversion of reactant R to product A is under control and is preferred at temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, high The conversion of reactant R to product B is under control and is preferred at temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, higharrow_forwardWhat's the mechanism for the following reaction scheme?arrow_forward
- Explain the mechanism of dissolving Metal Reduction of an Alkyne to a Trans Alkene ?arrow_forwardDetermine the mechanism of nucleophilic substitution for attached reaction anddraw the products, including stereochemistry.arrow_forwardWhich of the reactions in has the least kinetically stable product?arrow_forward
- How can you distinguish in a SNAr reactionwhether it will occur according to an addition- elimination or an elimination-addition mechanism?arrow_forwardQues Draw the mechanism for the reaction of methylcyclopentane with chlorine under UV light Include all possible free-radicals developed in the propagation steparrow_forwardWhich type of reaction - addition, elimination, rearrangement, substitution, - best describes each of the following?arrow_forward
- a) Consider the reaction of HBr with ethylene and propylene. At roomtemperature the reaction of propylene with HBr is much faster than thereaction with ethylene.Using reaction energy diagrams and your knowledge of carbocationstability explain why this is so. b) Xylene (dimethylbenzene) is a commonly used chemical in the printingindustry and as a cleaning solvent for oily waste. It is also used whenpreparing histological samples to remove waxes from biological samples.Draw the three possible structures for this compound and give the UPACnames for each. Define which structures are ortho, meta, and para.arrow_forwardat what condition we can isobutylene as an ideal gas?and why?arrow_forwardThis is a hydrogenation and carbene addition problem. How do I find what the missing reactant is of the reaction?arrow_forward


 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


