
Concept explainers
Interpretation:
The amino acid tyrosine is biologically degraded by a series of steps that include the following transformations:
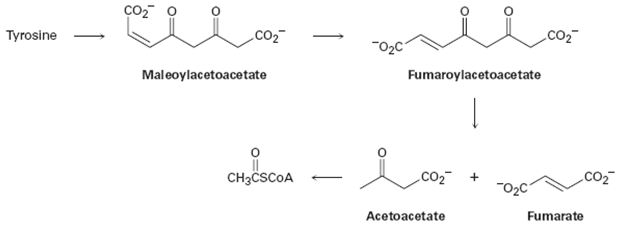
Concept introduction:
α-amino acid, in addition to their role as proton monomeric units, are energy
1) Essential amino acid
2) Non essential amino acid
While the non essential amino acid can be synchronized from metabolic processor, the essential amino acid must be provide by the diet. However, the essential dietary amino acid, instead of being extract or stored for future use, are converted to common metabolic intermediate like pyruvate, oxaloacetate, acetyl CoA, α-ketoglutarate etc. Thus amino acid are also precious of glucose, fatty acid, and
For the present, tyrosine clarifies itself to be a non essential amino acid, it is thus a metabolic processor and need not be supplied by the diet.
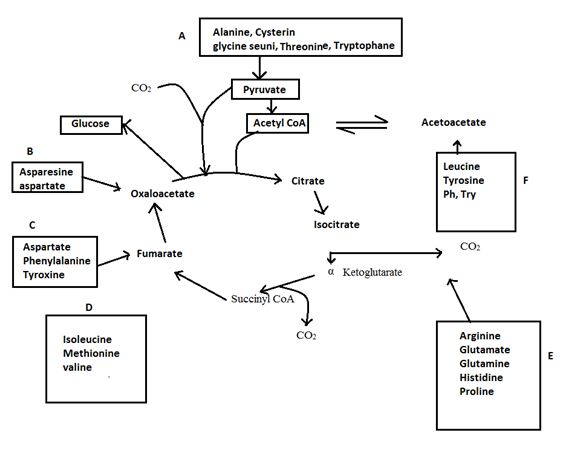
Further tyrisone is alos both a glucogenic and ketogenic amino acid. Its carbon skeleton can be degraded to either of pyruvate, α-ketoglutarate acetyl CoA, fumarate or oxaloacetate glucogenic amino acid and also to acetyl CoA or aceto acetate to be converted to ketone bodies or fatty acid. (ketogenic amino acid)
Degradation of amino acids to one of seven common metabolic intermediates
Want to see the full answer?
Check out a sample textbook solution
Chapter 29 Solutions
Organic Chemistry
- Identify the modes of catalysis and when / why they occur• Acid-base • Covalent• Transition state stabilization• Catalysis by proximity • Catalysis by strain• Lock & key is better seen as describing substrate binding to active sitearrow_forwardChemistry Dimethylphenyl-oxopropanoate decarboxylase catalyzes the following reaction. Draw the structure of the cofactor used by this enzyme and propose a mechanism for this reaction.arrow_forwardMany multi-step organic reaction mechanisms involve proton transfer steps. For example, the first step of Fischer esterification of carboxylic acids(as shown with acedic acid) is activation of the acid by protonation. Based on your understanding of which reaction pathway is more favorable, explain why using chemical structures.arrow_forward
- NAD is: B E) . Amines can be produced by: A) B) Oxidation of nitro compounds Reduction of nitro compounds Acidification of nitro compounds Hydrolysis of nitro compounds IUPAC name of the following compound is The reduced form of NADH A dehydrating coenzyme A hydrating coenzyme A methylating coenzyme The oxidized form of NADH A) 3-methylhexanoic acid 3-methylbenzoicacid 3-methylcyclohexanoic acid. 1-methylcyclohexanoic acid A) B) Peptide bonds are: A) B D) The compound 4-hydroxyhexanedioc acid has following functional groups (s) 2 Carboxylic acids alcohol and carboxylic acid ester and alcohol alcohol and aromatic group ක COOH Esters Amines Amides Carboxylic acids and amines CH₁ 3-metnyl кустоarrow_forward(a) What is the difference between the hormones progesterone and testosterone? (b) Draw the structure of a a steroid nucleus. (c) Give the products obtained from complete base hydrolysis in the following reaction: O || CH,−O−C−(CH2)14–CH3 O CH–0–C−(CH2)14—CH3 + 3 NaOH O CH,−0–C−(CH2)14–CH3arrow_forwardPlease draw by hand. Triosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme's catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a base and an acid catalyst, respectively. Propose a mechanism for the reaction. ОН 2-03Р0 ОН dihydroxyacetone phosphate triosephosphate isomerase 2-03РО. H glyceraldehyde-3-phosphate FYI Glu is glutamic acid and his is histadinearrow_forward
- α-Amino acids can be prepared by treating an aldehyde with ammonia/trace acid, followed by hydrogen cyanide, followed by acid-catalyzed hydrolysis. Draw the structures of the two intermediates formed in this reaction.arrow_forwardWhich of the following bases are strong enough to deprotonate C6H5OH(pKa = 10) so that equilibrium favors the products:(a) H2O; (b) NaOH; (c) NaNH2; (d) CH3NH2; (e) NaHCO3; (f) NaSH; (g)NaH?arrow_forwardTriosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme’s catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a general-base and a general-acid catalyst, respectively. Propose a mechanism for the reaction.arrow_forward
- Under acidic conditions (such as in your stomach) benzyl penicillin (BP) breaks down to three products which we will call A, B, and C in a parallel reaction with the following A BP B. values for the rate constants: ka = 7.0 × 10-4 sec-1; ks = 4.1 x 10-3 sec-1; and ke 5.7 x 10-3 sec-1. What is the yield of product A? In other words, after the reaction has reached completion what fraction of the product is present as compound-A?arrow_forward1 Norepinephrine can be metabolized to the following metabolite in two steps as shown below. What might be the enzymes (in the correct order) responsible for these biotransformations? но но но HN -OH но- но но Он OH OH norepinephrine COMT / MAO MAO / Cyp450 MAO / Aldehyde dehydrogenase Сур450 / МАОarrow_forwardThe rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M- 1s - 1. In the presence of Co2 +, the rate constant is 1.5 * 106 M- 1s - 1. What rate enhancement does the catalyst provide?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


