
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 29.SE, Problem 31MP
Interpretation Introduction
Interpretation:
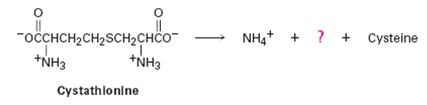
The amino acid cysteine, C3H7NO2S, is biosynthesized from a substance called cystathionine by a multistep pathway.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) What is the difference between the hormones progesterone and testosterone?
(b) Draw the structure of a a steroid nucleus.
(c) Give the products obtained from complete base hydrolysis in the following reaction:
O
||
CH,−O−C−(CH2)14–CH3
O
CH–0–C−(CH2)14—CH3 + 3 NaOH
O
CH,−0–C−(CH2)14–CH3
Determine which of the following bases is strong enough to deprotonate acetonitrile (CH3CN), so that equilibrium favors the products:(a) NaH; (b) Na2CO3; (c) NaOH; (d) NaNH2; (e) NaHCO3.
The first step in the catabolism of most amino acids is the removal of the nitrogen atom by transfer to an a-keto acid, a reaction
catalyzed by an enzyme called a transaminase. The a-keto acid acceptor is often a-ketoglutarate. Modify the structures in the
product to show the products of the transamination of cysteine. Be sure to show functional groups with the charge and number of
attached hydrogen atoms appropriate for pH 7.4.
transaminase
+
O=C
H₂N-CH
+
CH₂
CH₂
CH₂
SH
Incorrect
H₂N
||
CH
|
CH₂
|
CH₂
I
||
O
||
n
|
CH₂
T
SH
Chapter 29 Solutions
Organic Chemistry
Ch. 29.1 - Prob. 1PCh. 29.3 - Write the equations for the remaining passages of...Ch. 29.3 - Prob. 3PCh. 29.4 - Write a mechanism for the dehydration reaction of...Ch. 29.4 - Evidence for the role of acetate in fatty-acid...Ch. 29.4 - Does the reduction of acetoacetyl ACP in step 6...Ch. 29.5 - Prob. 7PCh. 29.5 - Look at the entire glycolysis pathway, and make a...Ch. 29.6 - Prob. 9PCh. 29.7 - Prob. 10P
Ch. 29.7 - Write mechanisms for step 2 of the citric acid...Ch. 29.7 - Prob. 12PCh. 29.8 - Prob. 13PCh. 29.9 - Write all the steps in the transamination reaction...Ch. 29.9 - What -keto acid is formed on transamination of...Ch. 29.9 - Prob. 16PCh. 29.SE - Prob. 17VCCh. 29.SE - Identify the following intermediate in the citric...Ch. 29.SE - The following compound is an intermediate in the...Ch. 29.SE - Prob. 20VCCh. 29.SE - In the pentose phosphate pathway for degrading...Ch. 29.SE - Prob. 22MPCh. 29.SE - One of the steps in the pentose phosphate pathway...Ch. 29.SE - One of the steps in the pentose phosphate pathway...Ch. 29.SE - Prob. 25MPCh. 29.SE - Prob. 26MPCh. 29.SE - Prob. 27MPCh. 29.SE - Prob. 28MPCh. 29.SE - Prob. 29MPCh. 29.SE - Prob. 30MPCh. 29.SE - Prob. 31MPCh. 29.SE - Prob. 32APCh. 29.SE - Prob. 33APCh. 29.SE - Prob. 34APCh. 29.SE - Prob. 35APCh. 29.SE - Prob. 36APCh. 29.SE - Prob. 37APCh. 29.SE - Prob. 38APCh. 29.SE - Prob. 39APCh. 29.SE - Prob. 40APCh. 29.SE - Prob. 41APCh. 29.SE - Prob. 42APCh. 29.SE - Prob. 43APCh. 29.SE - Prob. 44APCh. 29.SE - Prob. 45APCh. 29.SE - Prob. 46APCh. 29.SE - Prob. 47APCh. 29.SE - Prob. 48APCh. 29.SE - Prob. 49APCh. 29.SE - Prob. 50APCh. 29.SE - In glycerol metabolism, the oxidation of...Ch. 29.SE - Prob. 52APCh. 29.SE - Prob. 53APCh. 29.SE - Prob. 54APCh. 29.SE - In step 7 of fatty-acid biosynthesis (Figure...Ch. 29.SE - Prob. 56AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (a) Predict the product of the reaction of KOH with 1-amino propane. (b) Predict the product of a deprotonated ethanol (an “ethanolate anion", O-CH2-CH3) with phenol (hydroxybenzene). (c) Predict the product of propanoic acid with deprotonated ethanol (an “ethanolate anion", O-CH2-CH3).arrow_forwardFrom which amino acid are serotonin and melatonin synthesized? What types of reac- tions are involved in their biosynthesis? HO, CH,CH,NH, CH,O, CH,CH,NHCCH, (a) (b) H Serotonin Melatoninarrow_forwardPredict the products obtained from the reaction of triolein with the following reagents.(a) NaOH in water (b) H2 and a nickel catalyst (c) Br2 in CCl4arrow_forward
- Look up the structure of lisdexamfetamine (Vyvanse), a drug used in the treatment of attention deficit hyperactivity disorder (ADHD). Redraw it and identify all the functional groups present. What is known about itstherapeutic properties?arrow_forwardIf H218O is used in the hydrolysis reaction catalyzed by lysozyme, which ring will contain the label, NAMor NAG?arrow_forwardWhich of the following bases are strong enough to deprotonate C6H5OH(pKa = 10) so that equilibrium favors the products:(a) H2O; (b) NaOH; (c) NaNH2; (d) CH3NH2; (e) NaHCO3; (f) NaSH; (g)NaH?arrow_forward
- Draw the following compounds. a) (25,5Z)-2-amino-6-bromohept-5-enoic acid b) (S)-2,3-epoxy-4-hydroxycyclohexanonearrow_forwardWhich of the following bases is strong enough to deprotonate N,Ndimethylacetamide [CH3CON(CH3)2, pKa = 30], so that equilibrium favors the products: (a) NaNH2; (b) NaOH?arrow_forwardDetermining if a Base Is Strong Enough to Deprotonate an Acid Which of the following bases is strong enough to deprotonate N,Ndimethylacetamide [CH3CON(CH3)2, pKa = 30], so that equilibrium favors the products:(a)NaNH2; (b) NaOH?arrow_forward
- Indicate whether each statement is true or false. (a) Tryptophan is an aromatic amino acid. (b) Lysine is positively charged at pH 7. (c) Asparagine has two amide bonds. (d) Isoleucine and leucine are enantiomers. (e) Valine is probably more water-soluble than arginine.arrow_forward13. Of the listed drugs, the hydroxamic reaction gives: A) Calcium lactate B) Gamma-aminobutyric acid C) Piracetam D) Potassium acetatearrow_forwardWhen CaC2 reacts with water, what carbon-containing compoundforms?(a) CO (b) CO2 (c) CH4 (d) C2H2 (e) H2CO3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY