
Concept explainers
(a)
Interpretation:
The classification of the given molecule as achiral or chiral is to be stated.
Concept introduction:
A carbon atom that has four nonequivalent atoms or groups attached to it is known as the chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers. A carbon atom that does not have four nonequivalent atoms or groups attached to it is known as an achiral carbon atom.
Answer to Problem 6.1P
The given molecule is a chiral molecule.
Explanation of Solution
The structure of the given compound is shown below as,
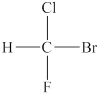
Figure 1
Four different groups are attached to the carbon atom that means it is a stereogenic center. The molecule in which stereogenic center is present is known as the chiral molecule. Therefore, the given molecule is a chiral molecule.
The given molecule is a chiral molecule.
(b)
Interpretation:
The classification of the given molecule as achiral or chiral is to be stated.
Concept introduction:
A carbon atom that has four nonequivalent atoms or groups attached to it is known as the chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers. A carbon atom that does not have four nonequivalent atoms or groups attached to it is known as an achiral carbon atom.
Answer to Problem 6.1P
The given molecule is an achiral molecule.
Explanation of Solution
The structure of the given compound is shown below as,
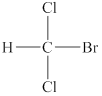
Figure 2
Two same groups are attached to the carbon atom that means it is not a stereogenic center. The molecule in which no stereogenic center is present is known as an achiral molecule. Therefore, the given molecule is an achiral molecule.
The given molecule is an achiral molecule.
(c)
Interpretation:
The classification of the given molecule as achiral or chiral is to be stated.
Concept introduction:
A carbon atom that has four nonequivalent atoms or groups attached to it is known as the chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers. A carbon atom that does not have four nonequivalent atoms or groups attached to it is known as an achiral carbon atom.
Answer to Problem 6.1P
The given molecule is a chiral molecule.
Explanation of Solution
The structure of the given compound is shown below as,
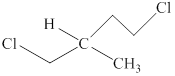
Figure 3
Four different groups are attached to the carbon atom that means it is a stereogenic center. The molecule in which stereogenic center is present is known as the chiral molecule. Therefore, the given molecule is a chiral molecule.
The given molecule is a chiral molecule.
(d)
Interpretation:
The classification of the given molecule as achiral or chiral is to be stated.
Concept introduction:
An atom that has four nonequivalent atoms or groups attached to it is known as the chiral atom. Chiral centers are also called as asymmetric or stereogenic centers. An atom that does not have four nonequivalent atoms or groups attached to it is known as an achiral atom.
Answer to Problem 6.1P
The given molecule is a chiral molecule.
Explanation of Solution
The structure of the given compound is shown below as,
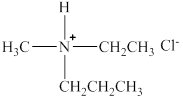
Figure 4
Four different groups are attached to the nitrogen atom that means it is a stereogenic center. The molecule in which stereogenic center is present is known as the chiral molecule. Therefore, the given molecule is a chiral molecule.
The given molecule is a chiral molecule.
Want to see more full solutions like this?
Chapter 6 Solutions
Organic Chemistry
- which one of the following cycloalkanes will be least stable? (a) cyclopropane (b) cyclobutane (c) cyclopentan (d) cyclohexanearrow_forward(a) Which compounds (B–F) are identical to A? (b) Which compounds (B–F) represent an isomer of A?arrow_forwardDraw the more stable chair conformation for each of the following disubstituted cyclohexanes. (a) OH (b) OH (c) (d) (e) (f) „CBr3 CBr3 "Cl3 CCI3 Cl3arrow_forward
- Name the following structures. (a) (b) (c) CI ÇIarrow_forward(B) Draw two chair conformations of the following substituted cyclohexane and identify the most stable conformation. Clearly show the axial and equatorial bonds.arrow_forward(a) which if the structure of trans-1,2-dimethylcyclopentane? (b) which is the most stable conformation of 1-bromo-2-ethylcyclohexane? (c) which is the least stable conformation of 1-bromo-2-ethylcyclohexane? (d) which is the more stable configuration of 1,3-dimethylcyclopentane? *Et = ethylarrow_forward
- Which molecule is expected to show aromatic behavior (the true geometries are flexible)? (a) (b) (c) (d)arrow_forwardDraw a bond-line diagram for the compounds below. Which of the following compounds is chiral? (a) 2-bromobutane (b) methylcyclohexane (draw a chair configuration)arrow_forwardWhich do you expect to be the more stable conformation of cis-1,3-dimethylcyclobutane, A or B? Why?arrow_forward
- Draw the more stable chair conformation of each of the following compounds in which an sp²-hybridized carbon atom is part of the ring. (a) (b) (c) (d) (e) Brarrow_forwardFor each of the following compounds, identify all groups that would be considered substituents and then indicate the systematic name for each compound. (a) (b) (d) (e)arrow_forwardFollowing are the alternative chair conformations for trans-1,2-dimethylcyclohexane.(a) Estimate the difference in free energy between these two conformations.(b) Given your value in (a), calculate the percent of each chair present in anequilibrium mixture of the two at 25°C.arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
