
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.2, Problem 6.3P
For each of the following compounds,
- A. give the IUPAC name
- B. give the common name (if possible).
- C. classify the compound as a methyl, primary, secondary, or tertiar
- a. (CH3)2CHCH2C1
- b. (CH3)3CBr
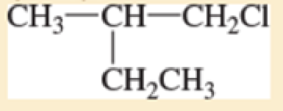
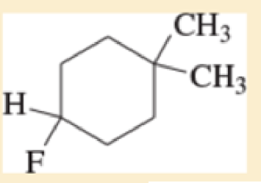
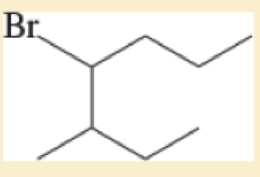
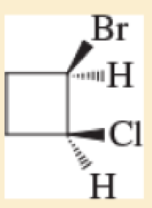
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are the systematic (IUPAC) names for the compounds shown?
A.
OH
H3C
systematic (IUPAC) name:
B. H3C-CH₂-CH-CH₂
LL
CH3 OH
systematic (IUPAC) name:
A. Give the IUPAC and Common names the following compounds.
1.
CH3CH,OCH(CH3)2
4.
H
CH,CH;
CH,CH,
`H.
5.
CH3
OCH3
CH3
3.
OCH,CH3
6.
2.
Acetophenone
Condensed Structure:
Product formed:
A.Oxidation (K2Cr2O7/H2SO4)
b.Reduction (NaBH4/H2O)
c. Addition of alcohol ( CH3OH) → with another addition of alcohol (C2H5OH)
Chapter 6 Solutions
Organic Chemistry (9th Edition)
Ch. 6.1 - Classify each compound as an alkyl halide, a vinyl...Ch. 6.2 - Give the structures of the following compounds. a....Ch. 6.2 - For each of the following compounds, A. give the...Ch. 6.3E - Prob. 6.4PCh. 6.4 - Prob. 6.5PCh. 6.5A - For each pair of compounds, predict which compound...Ch. 6.5B - Prob. 6.7PCh. 6.6B - Prob. 6.8PCh. 6.6B - The light-initiated reaction of...Ch. 6.6B - Show how free-radical halogenation might be used...
Ch. 6.7 - Prob. 6.11PCh. 6.7 - Prob. 6.12PCh. 6.8 - Prob. 6.13PCh. 6.9 - Predict the major products of the following...Ch. 6.9 - Prob. 6.15PCh. 6.10A - Prob. 6.16PCh. 6.11A - When diethyl ether (CH3CH2OCH2CH3) is treated with...Ch. 6.11B - Prob. 6.18PCh. 6.11B - For each pair of compounds, state which compound...Ch. 6.12 - Prob. 6.20PCh. 6.12 - Under appropriate conditions...Ch. 6.13 - Propose an SN1 mechanism for the solvolysis of...Ch. 6.13B - Prob. 6.23PCh. 6.13B - 3-Bromocyclohexene is a secondary halide, and...Ch. 6.15 - Prob. 6.25PCh. 6.15 - Prob. 6.26PCh. 6.16 - For each reaction, give the expected substitution...Ch. 6.16 - Prob. 6.28PCh. 6.16 - Prob. 6.29PCh. 6 - Prob. 6.30SPCh. 6 - Draw the structures of the following compounds. a....Ch. 6 - Give systematic (IUPAC) names for the following...Ch. 6 - Prob. 6.33SPCh. 6 - Predict the compound in each pair that will...Ch. 6 - Prob. 6.35SPCh. 6 - Give two syntheses for (CH3)2CHOCH2CH3, and...Ch. 6 - Prob. 6.37SPCh. 6 - Prob. 6.38SPCh. 6 - Chlorocyclohexane reacts with sodium cyanide...Ch. 6 - Give the substitution products expected from...Ch. 6 - Prob. 6.41SPCh. 6 - Prob. 6.42SPCh. 6 - Two of the carbocations in Problem6-42 are prone...Ch. 6 - Prob. 6.44SPCh. 6 - Predict the products of the following SN2...Ch. 6 - Prob. 6.46SPCh. 6 - Strawberry growers have used large quantities of...Ch. 6 - A solution of pure (S)-2-iodobutane ([]=+15.90) in...Ch. 6 - Prob. 6.49SPCh. 6 - Give a mechanism to explain the two products...Ch. 6 - Prob. 6.51SPCh. 6 - Because the SN1 reaction goes through a flat...Ch. 6 - Prob. 6.53SPCh. 6 - Furfuryl chloride can undergo substitution by both...Ch. 6 - Prob. 6.55SPCh. 6 - The following reaction takes place under...Ch. 6 - Propose mechanisms to account for the observed...Ch. 6 - Prob. 6.58SPCh. 6 - Prob. 6.59SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, & Biological Chemistry
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
Give the IUPAC name for each compound.
Organic Chemistry
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- B. Give the IUPAC name for each compound. F CH3 ÇI 1. CH;CH2CHCH2CH2CHCH3 5. CH,CH3 F 2. CI 6. OH CI NO, 7. NO, NO, Br 8. Br 3.arrow_forward4. Use the IUPAC Nomenclature System to name each of the following compounds: a. b. CH,CCH₂CH, 0 HCCHCH₂CH₂ CI O -C-C-CH₂ Br CHỊCH,CH,CH, CH,CCH₂CH₂CH CH, CI CH₂CHCH₂CH Br CH, Ô CH,CCH.CCH.CH.CH, OH 0 CH₂ CH,CCH₂CH.CH,CHCH,C-H H,CHCH.CH CH₂arrow_forwardInstructions: Give the IUPAC name for each compound. a. CH3CHCH₂CH₂CH₂CH3 Br Instructions: DRAW the structure corresponding to each name a. 3-chloro-2-methylhexane b. 4-ethyl-5-iodo-2,2-dimethyloctane c. 1,1,3-tribromocyclohexane d. propyl chloride b. (CH3)2CHCHCH₂CH3 CI Instructions: Give the IUPAC name for each thiol. a. CH3CH₂CHCH₂CH3 SH b. SH CH3 'Br C. (CH3CH₂)2CHCH₂CH₂CH₂CH₂SHarrow_forward
- A. Naming Structures 1. Use the JUPAC rules to name the following structures. CH CH = CHCHCH, b. CH3 a. CH3 CH2 CH3 CH-CH сн, CH3CHCH2CH =CHCCH, CH3 2. Use IUPAC rules to name the following structures. CH,CH3 b. CH3 CH3 CH3 CH,CH, CH3CH, В. B. Draw the structures of the following cycloalkanes 1. 2,3-dimethyl-5-propyldecane 2. 3,4,5-triethyloctane 3. 1-ethyl-3-propylcyclopentane 4. 1,2,2,4-tetramethylcyclohexanearrow_forward1. Provide the IUPAC name for each of the following organic molecules. a. b. C. H3C H3C, Home H3C CH3 CH3 CH3 Br H 2. Draw structures for the following organic molecules, using the wedged/hashed line convention to depict any stereochemical relationships between groups: a. cis-3-ethylcyclobutane-1-carbaldehyde b. (R)-1-chloro-6-iodo-6-methyloctan-3-onearrow_forwardWhat is the IUPAC name for the following compound? CH₂CH₂ C CH3-CH₂-CH-CH₂-CH-CH-CH-CH₂ CI CH3 Select one: O A. 3,7-dichloro-5-ethyl-6-methyloctane O B. 2-chloro-3-methyl-4-(2-chlorobutyl) hexane O C. 2,6-dichloro-4-ethyl-3-methyloctane O D. 3-chloro-5-(2-chloro-1-methylpropyl) heptane OE. 2,3,4,6-dichloroethylmethyoctanearrow_forward
- Give the IUPAC name for the following compound. H H₂C H H CH3 CH-CH,-CH,-CH3 CH H3C CH3 Select one: O A. 3-methyl-5-isopropylheptane OB. 2-methyl-4-isopropylheptane OC. 2-methyl-4-isopropyloctane O D. 3-methyl-5-isopropylhexanearrow_forward1. Provide proper IUPAC names for the following compounds. a. CH3 | CH3C CCHCH2CH2CH3 CH3 Br b. H₂C=CH-C-CH-CC-H | CH3 C.arrow_forward#544 of Isomers (chiral) Which of the following 2-chloro-1-butanol (A), 3-chloro-1-butanol (B), and 4-chloro-1-butanol (C), contain a stereogenic carbon atom. Select one: a. A only O b. B only O c. C only O d. A and B e. A, B and Carrow_forward
- 8. Name the following compounds using IUPAC name (common names are not allowed for example, Isopropyl, tertiary butyl, sec-butyl, etc). thu a) b) d) CH3CH₂C(CH3)2CH₂CH₂CH(CH₂CH3)C(CIBr)CH3 C)arrow_forwardGive the IUPAC name of the structure below: a.trans-2-methyl-2-en-4-propyl-hept-6-yne b.trans-4-propyl-5-en-hepty-1-yne c.cis-4-butyl-1-yne-octy-5-ene d.cis-2-methyl-2-en-4-propyl-hept-6-ynearrow_forward4. Syn and anti addition can occur with alkenes. 1. Describe syn and anti addition. b. Give an example of syn addition to an alkene and explain why it is syn. c. Give an example of anti addition to an alkene and explain why it is anti. 5. The following reactions shows the dehydration of 2-methylcyclohexanol. Why is 1-methylcyclohexene the major product? X OH H3PO4 84% 16%arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY