
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Question
Chapter 9, Problem 16Q
Interpretation Introduction
Interpretation:
Taking 10 plastic containers of one’s choice it should be identified that which polymer is most frequently encountered in the sample.
Concept Introduction:
Monomer: A molecule is considered as monomer when this molecule bonds with another identical molecule which results to form polymer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
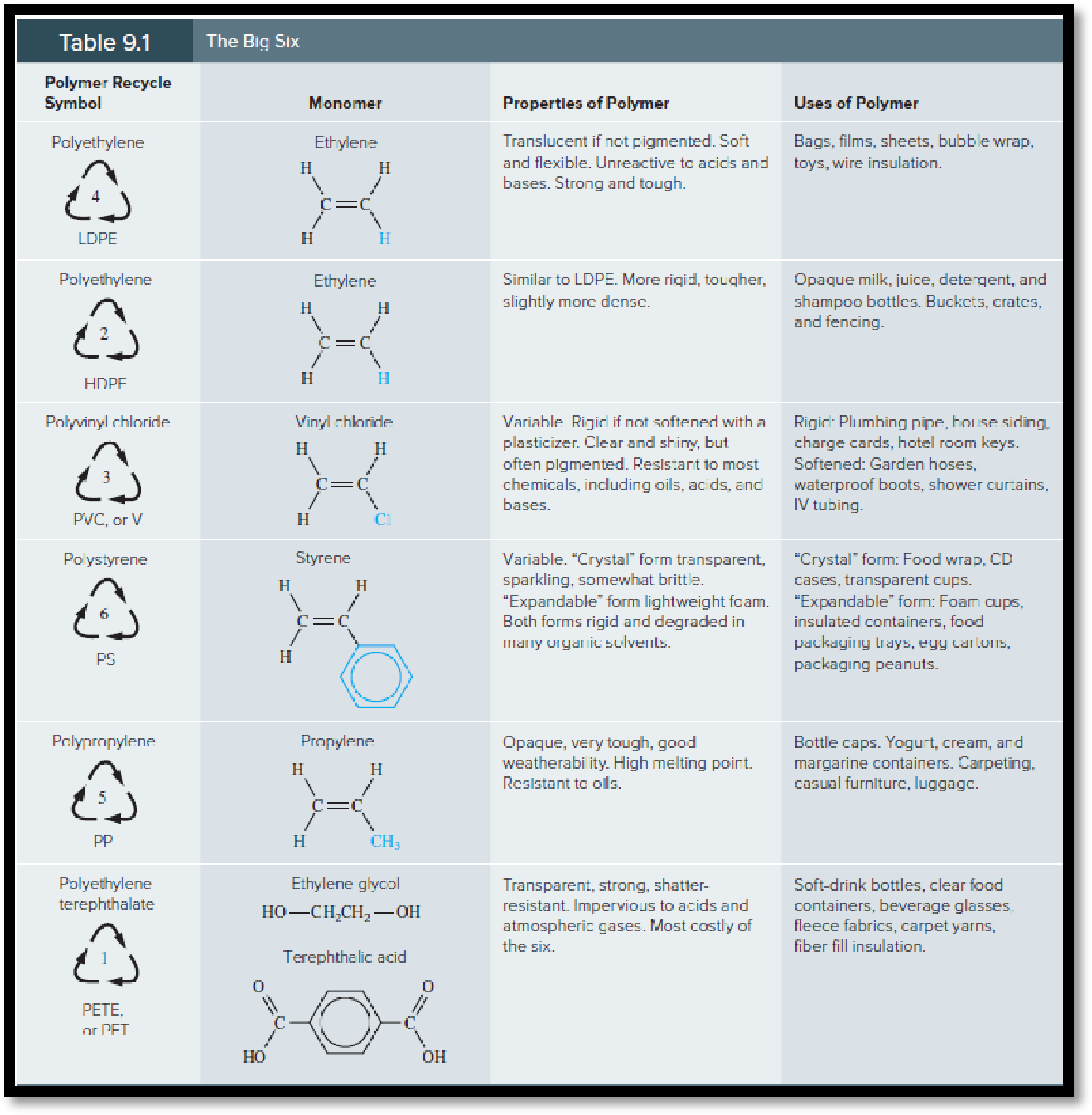
Paragrph 4: Discusses the importance of
recycling polymers and includes a recycling chart
that list the 7 recycles codes and the polymer
type.
Define Polymers and Polymerization ?
A polymer synthesis method wherein a bulk material is subjected to fabrication and printing of desired nano shapes by removing a portion of a material.
a. nanolithography
b. Sol-gel
c. Pyrolysis
d. sputtering
Chapter 9 Solutions
Chemistry In Context
Ch. 9.1 - Scientific Practices Tennis Anyone? Examine this...Ch. 9.3 - Prob. 9.2YTCh. 9.3 - Prob. 9.3YTCh. 9.4 - Prob. 9.4YTCh. 9.4 - Prob. 9.5YTCh. 9.4 - Prob. 9.6YTCh. 9.4 - Prob. 9.7YTCh. 9.4 - Prob. 9.8YTCh. 9.4 - Prob. 9.9YTCh. 9.5 - Prob. 9.10YT
Ch. 9.5 - Skill Building Benzene and Phenyl The difference...Ch. 9.5 - Prob. 9.13YTCh. 9.5 - Skill Building Polystyrene Possibilities Show the...Ch. 9.6 - Skill Building Esters and Polyesters You have seen...Ch. 9.6 - Prob. 9.16YTCh. 9.7 - Skill Building Kevlar Kevlar is a polyamide used...Ch. 9.8 - Prob. 9.20YTCh. 9.8 - Your Turn 9.22 Skill Building Burning a Plastic...Ch. 9.8 - Your Turn 9.23 Scientific Practices Landfill...Ch. 9.9 - Examine the values in Table 9.4 from the American...Ch. 9.9 - Prob. 9.25YTCh. 9.9 - Prob. 9.26YTCh. 9.9 - Prob. 9.28YTCh. 9.10 - Skill Building The Chemistry of PLA We dont show...Ch. 9.11 - Your Turn 9.31 Scientific Practices Glass or...Ch. 9.11 - Prob. 9.32YTCh. 9.11 - Skill Building Meet DEHP DEHP belongs to a common...Ch. 9 - Prob. 1QCh. 9 - Prob. 2QCh. 9 - Prob. 3QCh. 9 - Prob. 4QCh. 9 - Prob. 5QCh. 9 - Prob. 6QCh. 9 - Prob. 7QCh. 9 - Prob. 8QCh. 9 - Prob. 9QCh. 9 - Prob. 10QCh. 9 - Prob. 11QCh. 9 - Prob. 12QCh. 9 - Prob. 13QCh. 9 - Prob. 14QCh. 9 - Prob. 15QCh. 9 - Prob. 16QCh. 9 - Prob. 17QCh. 9 - Prob. 18QCh. 9 - Prob. 19QCh. 9 - Prob. 20QCh. 9 - Prob. 21QCh. 9 - Prob. 22QCh. 9 - Prob. 23QCh. 9 - Prob. 24QCh. 9 - Prob. 25QCh. 9 - Prob. 26QCh. 9 - Prob. 27QCh. 9 - Prob. 28QCh. 9 - Prob. 29QCh. 9 - Prob. 30QCh. 9 - Prob. 31QCh. 9 - Prob. 32QCh. 9 - Prob. 33QCh. 9 - Prob. 34QCh. 9 - Prob. 35QCh. 9 - Prob. 36QCh. 9 - Prob. 37QCh. 9 - Prob. 38QCh. 9 - Prob. 39QCh. 9 - Prob. 40QCh. 9 - Prob. 41QCh. 9 - Prob. 42QCh. 9 - Prob. 43QCh. 9 - Prob. 44QCh. 9 - Prob. 45QCh. 9 - Prob. 46QCh. 9 - Prob. 47QCh. 9 - Prob. 48QCh. 9 - Prob. 49QCh. 9 - Prob. 50QCh. 9 - Prob. 51QCh. 9 - Prob. 52QCh. 9 - Prob. 53QCh. 9 - Prob. 54QCh. 9 - Prob. 55QCh. 9 - Prob. 56QCh. 9 - Prob. 57QCh. 9 - Prob. 58QCh. 9 - Prob. 59Q
Knowledge Booster
Similar questions
- Use the web to research the amount of PVC polymer produced annually in the United States. What are the three most common uses of this polymer?arrow_forwardDiscuss some of the natural polymers? What is their role?arrow_forward6. Polyethene, aka Polyethylene, is what type of polymer? a. Addition b. Condensation C. Polyamide d. Polyesterarrow_forward
- What are the applications of polymers in real life?arrow_forwardWhich one of the following cannot be used as a deodorant? 2. a. CaCo3 b. Zn(C17H35CO0)2 C. Al(OH)3 d. Zno Which one of the following is not a suitable disinfectant against corona virus? a. Ethanol solution b. NaAl(OH)4 solution c. Phenol solution d. NaCIO solution Which one of the following phrases is not true? a. Acidic media do not affect soapless soaps b. Synthetic detergents are stronger than soaps C. Synthetic detergents cannot be used in hard water d. Alkylbentene sulphonates are synthetic detergents Dettol is: a. Antifertility. b. Antis ptic. C. Antipyretic. d. Antibiotic. Which one of the following is not an antacid? a. Mg(OH)2 b. CH3COONa c. Milk d. NaHCO3arrow_forwardReferences] VISUALIZATION Polyester Formation II HO-A-OH HO-C-B-C-0H ethylene glycol (A group) terephthalic acid (B group) The starting materials for this reaction are ethylene glycol and terephthalic acid. The letters A and B represent organic groups that are unreactive. What reactive organic functional group does ethylene glycol (containing the A functional group) contain? Enter its name. Check (1 of 7)arrow_forward
- 7. Statement 1: All food additives are carcinogenic. Statement 2: Food additives must be avoided as much as possible. A. True, False B. True, True C. False, False D. False, True 8. How do thickeners improve a food product? A. They prevent spoilage due to oxygenation B. They increase the viscosity of a product C. They preserve the flavor of a product D. They keep food from drying out. _9. It makes the food thicker by keeping oil and water mixed together. A. Emulsifiers C. Humectants B. Flavor Enhancer D. Antioxidantsarrow_forwardنقطة واحدة نقطة واحدة نقطة واحدة نقطة واحدة = ||| can be heating and Cannot be heating The are .33 .reshaped is A. Thermoset. B. Thermoplastic. .34 .and reshaped is A. Thermoset. B. Thermoplastic. The polymer is if heated too.35 .much, they burn is A. Thermoset. B. Thermoplastic. O Production of .36 .plastic products A. Seven. B. Four. C. Three.arrow_forwardsearch for this information using the chemicals used in veterinary medicine that are in the image: 1. preliminary actions. If there is no information on preliminary actions, indicate that it does not apply. 2. Description of the substance disposal procedure (or correct use and handling of the equipment), here you must write: How am I going to get rid of the substance?, how to discard the substance, once we are done with it * search the MSDS and reliable sources for this informationarrow_forward
- Differentiate between thermoplastic and thermosetting polymers. Give one example of each.arrow_forward8. In a reduction reaction a. a carbon will form fewer bonds to carbon. b. a carbon will form fewer bonds with oxygen or more bonds to hydrogen. c a carbon will form more bonds with oxygen or fewer to hydrogen. d. two small molecules combine to form a large molecule. e. two large molecules react to form a large molecule and a small molecule. 9. What is an addition reaction? a. One in which atoms or groups of atoms are removed from an organic molecule and a double bond is formed. b. One in which an atom or small group replaces another atom or small group on the organic molecule. c. One in which atoms from a small molecule react with a double or triple bond in an organic molecule to become part of the molecule. d. One in which two large molecules combine to form one larger molecule and one small molecule, usually water. 10. What results when a secondary alcohol is oxidized? a. A ketone b. An amine c. An aldehyde d. An acid e. No reaction 11. Which type of reaction will an alkene not…arrow_forwardEXPLAIN HOW POLYCARBONATE WILL BE RECYCLED, MIXED, AND MOLDED AGAIN TO CREATE THE DESIRED SHAPEarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning  Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning