
a)
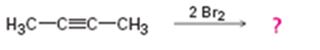
Interpretation:
Assuming that halogens add to
Concept introduction:
Alkynes when treated with one equivalent of a halogen yield a dihaloalkene as the product. They react with two equivalents of the halogens to yield a tetrahaloalkane derivative. In the first step of the addition reaction the nucleophilic attack of the π electrons of the double/triple bond in alkene/alkyne on a halogen results in the formation of a cyclic halonium ion with the simultaneous elimination of a halide ion. In the second step the halide ion attacks the cyclic halonium ion to yield the product.
To propose:
A mechanism and to predict the product(s) expected for the reaction in which two equivalents of Br2 adds to 2-butyne assuming that bromine adds to alkynes in the same manner as they add to alkenes.
b)
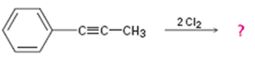
Interpretation:
Assuming that halogens add to alkynes in the same manner as they add to alkenes, a mechanism is to be proposed and the product(s) expected for the reaction in which two equivalents of Cl2 adds to 1-phenylpropyne is/are to be predicted.
Concept introduction:
Alkynes when treated with one equivalent of a halogen yield a dihaloalkene as the product. They react with two equivalents of the reagents to yield a tetrahaloalkane as the product. In the first step of the addition reaction, the nucleophilic attack of the π electrons of the double/triple bond in alkene/alkyne on a halogen results in the formation of a cyclic halonium ion with the simultaneous elimination of a halide ion. In the second step the halide ion attacks the cyclic halonium ion to yield the product.
To propose:
A mechanism and to predict the product(s) expected for the reaction in which two equivalents of Cl2 adds to 1-phenylpropyne assuming that chlorine adds to alkynes in the same manner as they add to alkenes.
c)
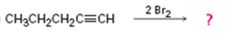
Interpretation:
Assuming that halogens add to alkynes in the same manner as they add to alkenes, a mechanism is to be proposed and the product(s) expected for the reaction in which two equivalents of Br2 adds to 1-pentyne is/are to be predicted.
Concept introduction:
Alkynes when treated with one equivalent of a halogen yield a dihaloalkene as the product. They react with two equivalents of the reagents to yield a tetrahaloalkane as the product. In the first step of the addition reaction the nucleophilic attack of the π electrons of the double/triple bond in alkene/alkyne on a halogen results in the formation of a cyclic halonium ion with the simultaneous elimination of a halide ion. In the second step the halide ion attacks the cyclic halonium ion to yield the product.
To propose:
A mechanism and to predict the product(s) expected for the reaction in which two equivalents of Br2 adds to 1-pentyne assuming that bromine adds to alkynes in the same manner as they add to alkenes.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- The addition of water to aldehydes and ketones occurs rapidly, although it is not thermodynamically favored. What would be the product for the reaction above? Hint: Think of the self-ionization of water and the polarity of the carbonyl group.arrow_forwardHow can alkenes and alkynes be classified: as nucleophiles, as electrophiles or depending on the reaction conditions could be both? Explain extensively.arrow_forwardExplain why the following alcohols, when heated with acid, form the same alkene.arrow_forward
- Draw the structure of the expected organic product(s) formed in the following reactions including correct stereochemistry. If the product is racemic write racemic or draw both isomers. Assume all reagents listed are present in excess unless otherwise noted. If no reaction occurs, state "No Reaction''.arrow_forwardShown below is a carbocation intermediate in an electrophilic addition reaction of HCl with two different alkenes. Draw structural formulas for both of the alkenes.arrow_forwardDraw the structure of the expected organic product formed in the following reactions including correct stereochemistry. If the product is racemic indicate this by either drawing both isomers or drawing one and writing racemic. Assume all reagent are present in excess unless otherwise noted. If no reaction occurs, state "No Reaction".arrow_forward
- The rate law for addition of Br2 to an alkenes is first order in Br2 and first order in the alkene. Does this information suggest that the mechanism of addition of Br2 to an alkene proceeds in the same matter as for addition of HBr? Explain.arrow_forwardAn isomer of tetramethylbenzene undergoes nitration to yield a single product. Based on this information, which isomer(s) of tetramethylbenzene could the starting material have been?arrow_forwardDraw the structures of two alkenes that would react to form the haloalkane below upon addition of HBr. Your alkenes should be different regioisomers that yield the haloalkane as the major product without requiring rearrangement to occur.arrow_forward
- Haloalkanes undergo nucleophilic substitution whereas haloarenes undergo electrophilic substitution. Explain.arrow_forwardSynthesize this molecule by using the principles of radical reactions, substitution, addition, and elimination reactions.Start with ethyne.arrow_forwardThe Friedel-Crafts Alkylation of p-xylene n-propyl bromide can result in an isopropyl as well as n-propyl substitution. A) Write the chemical equation involved in this reaction. B) Which will be the major and minor product? What do you expect will be the ratio of n-propyl to isopropyl substitution for p-xylene?arrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


