
Concept explainers
The skeleton is benzaldoxime is
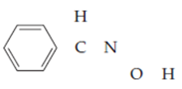
The number of valence electrons is as follows: from the phenyl group. ___; from each of two hydrogens, ___; from the carbon atom, ___; from the nitrogen atom, ___; and from the oxygen atom, ___, for a total of ___. Filling in the skeleton with single bonds and adding the appropriate unshared pairs gives
_____________________
The number of electrons in the
_____________________
in which carbon, nitrogen, and oxygen have their customary valences of ___, ___, and respectively. The alternative structure with a double bond between nitrogen and oxygen is
_____________________
This structure is not acceptable because it requires carbon and oxygen to exhibit the unfamiliar valences of ___ and ___.
Derive Lewis structures for the compounds below.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Pushing Electrons
- Among the four alkanes, ethane, propane, butane, and pentane, which is capable of existing in isomeric forms?arrow_forwardTotal: Complete the following questions, clearly and neatly showing all your work. 1. Give the name of the functional group present in each of the following molecules: H2N .N. ОН HO ОН 2. From the molecules shown above, give the formal chemical name of the one that is... a. ..most basic. b. ...most acidic. С. ...most likely to smell like fruit.arrow_forwardBitumen (tar) is a mixture of various unsaturated (and also saturated) hydrocarbons of different chain lengths (it is used to make road pavement - asphalt). Based on your correlation, suggest a physical explanation of the fact that tar is black (i.e. absorbs light of all wavelengths).arrow_forward
- An alkane, P, has the molecular formula, C,H.. An alkene, Q, has the molecular formula, C H,. (a) Name P and Q ánd write their full structural formulae. (b) State two differences between P and Q in terms of their structures. x'arrow_forwardGive the structural, abbreviated structural and skeletal formulas for the linear hydrocarbon with the molecular formula C4H10. You should either use the 'Building organic molecules' tool in Activity 1.1 of Topic 9 to draw the structural formula of the compound, or take a photograph of a hand-drawn structural formula that you have produced.arrow_forwardcomplete the following table & identify the 2 isomeric pairs among the structures.arrow_forward
- Which of these is a hydroxyl group? A large organic molecule with different functional groups. A group marked A consists of a nitrogen atom and two hydrogen atoms attached to it. A group marked B consists of a phosphorus atom with four oxygen atoms. One of these oxygen atoms is connected to the phosphorus by a double bond. All other bonds are single. Two oxygen atoms have negative charges. A group marked C consists of a carbon atom connected to an oxygen atom by a double bond. A group marked D consists of an oxygen atom connected to a hydrogen atom. The group marked E consists of a sulfur atom connected with a hydrogen atom. A group marked F consists of a carbon atom, which is connected to one oxygen atom by a double bond and the other oxygen atom by a single bond. This last mentioned oxygen atom also has a hydrogen attached. EDACFarrow_forwardFor each of the following hydrocarbons, provide the complete structural formula based on the name or abbreviated formula given. Determine the class or type of hydrocarbon (e.g. alkane, cycloalkane, alkene, alkyne, aromatic). Build the model for each.arrow_forwardThe skeleton of benzyldimethylamine is The number of available valence electrons is as follows: from the benzyl group, ___; from each of two methyl groups, ___; and, from the nitrogen atom, ___, for a total of ___. Filling in the skeleton with single bonds gives ____________________________ and adding the appropriate unshared pair gives ____________________________ The number of electrons in the functional group of this structure is ___, and the structure is (correct, incorrect).arrow_forward
- A. Structural Isomerism of Alkanes, continued. Construct models for the five alkanes that have the molecular formula C6H₁4. All five of your models should have the same number of each type of atom, but they should have the atoms connected in a different order. Thus the molecules they represent are structural isomers of one another. Note the tetrahedral geometry of each carbon atom. Draw an extended and a condensed structure (NOT skeletal/line angle) of each structural isomer, then determine its IUPAC name. Recall that since the IUPAC name specifies the number of each type of atom and how they are connected, each structural isomer will have a different name. Each name should very specifically describe the structure. Isomer 3: Extended Structure Isomer 3: Condensed Structure IUPAC Name: IUPAC Name: Isomer 4: Extended Structure Isomer 4: Condensed Structurearrow_forwardWill the number of isomers increase or decrease with thenumber of carbon atoms in a hydrocarbon? Explain yourreasoning.arrow_forwardwrite the chemical formula for each and determine the class of compound of each of the structuresarrow_forward

 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER


