
Pushing Electrons
4th Edition
ISBN: 9781133951889
Author: Weeks, Daniel P.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 7EQ
Dimethyl ether
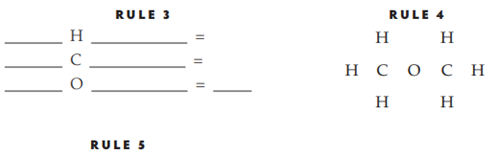
No. of electrons in structure _____
No. of valence electrons _____
Structure is ___________ (correct, incorrect)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Two major contributing resonance structures are possible for the anion shown. One is given, but is incomplete. Complete the
given structure by adding non-bonding electrons and formal charges. Draw the remaining structure, including non-bonding
electrons and formal charges. Omit curved arrows.
Draw formal charges and lone pairs to structure 1.
H
H
H
I.
с
|
H
H
Complete structure 2. Add the pi bond, lone pairs
and charges.
H
H
H
1.
A
|
1
H
H
H
compound
hydrogen-bonding force
Between
Between molecules of
formula or Lewis
molecules of the
the compound and
molecules of water?
name
structure
compound?
H
H
|
Н — С
|
|
yes
yes
N-chloromethylamine
-
no
no
H.
yes
yes
hydrogen iodide
HI
no
no
:0:
||
н —с —с-о — н
H
yes
yes
acetic acid
no
no
H
O O
Draw the Lewis Structure for C*H_{2} * S . How many double bonds are present?
Chapter 1 Solutions
Pushing Electrons
Ch. 1 - 1. Hydrogen is a Group I element and each...Ch. 1 - Methanol has the molecular formula CH4O. Its...Ch. 1 - 3. The skeleton of chloromethane is...Ch. 1 - 4. Methanol’s skeleton is
Connecting all bonded...Ch. 1 - 5. The structure for chloromethane is
It...Ch. 1 - Prob. 6EQCh. 1 - 7. Dimethyl ether
No. of electrons in...Ch. 1 - Methylamine (CH5N) No. of electrons in structure...Ch. 1 - Methanethiol (CH4S) No. of electrons in structure...Ch. 1 - Methylal (C3H8O2) No. of electrons in structure...
Ch. 1 - Prob. 11EQCh. 1 - Adding electrons to the skeleton by making single...Ch. 1 - This is done by removing an unshared pair from...Ch. 1 - Prob. 14EQCh. 1 - Prob. 15EQCh. 1 - Prob. 16EQCh. 1 - The skeleton of acetyl chloride is . Write the...Ch. 1 - Three constitutional isomers exist for the formula...Ch. 1 - A number of constitutional isomers exist for the...Ch. 1 - Using the method outlined above, derive the...Ch. 1 - Prob. 21EQCh. 1 - Prob. 22EQCh. 1 - Prob. 23EQCh. 1 - Prob. 24EQCh. 1 - The skeleton of benzyldimethylamine is
The...Ch. 1 - The skeleton is benzaldoxime is The number of...Ch. 1 - Prob. 27EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - Prob. 29EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - Prob. 31EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - The Lewis structure of acetone is Circling the...Ch. 1 - Chloromethane has the Lewis...Ch. 1 - In the Lewis structure for chloromethane, the...Ch. 1 - Prob. 36EQCh. 1 - The oxygen atom in acetone possesses ____ unshared...Ch. 1 - Nitrobenzene has the skeleton
The number of...Ch. 1 - Prob. 39EQCh. 1 - Compute and add on the formal charges I these...Ch. 1 - Prob. 41EQCh. 1 - Prob. 42EQCh. 1 - Prob. 43EQCh. 1 - Prob. 44EQCh. 1 - Prob. 45EQCh. 1 - Prob. 46EQCh. 1 - Prob. 47EQCh. 1 - Compute and add on the formal charges in these...Ch. 1 - Prob. 49EQCh. 1 - Prob. 50EQCh. 1 - The n-propyl cation can be formed from a molecule...Ch. 1 - Prob. 52EQCh. 1 - Prob. 53EQCh. 1 - Methanol, CH3OH, is a compound in which the formal...Ch. 1 - When a proton becomes bonded to diethyl ether, by...Ch. 1 - Tetrahydrofuran has the structure
When a proton...Ch. 1 - Prob. 57EQCh. 1 - Prob. 58EQCh. 1 - The structure of pyridine is
When a proton...Ch. 1 - The carbon atom owns one electron from each of ...Ch. 1 - The n-butyl anion can be formed from When the CLi...Ch. 1 - The isobutyl anion can be formed from When the CNa...Ch. 1 - Prob. 63EQCh. 1 - Ethanol, , is a compound in which the formal...Ch. 1 - The loss of a proton attached to the oxygen atom...Ch. 1 - A very strong base can remove a proton from...Ch. 1 - Prob. 67EQCh. 1 - Prob. 68EQCh. 1 - Prob. 69EQCh. 1 - The homolysis of the OO bond in diacetyl peroxide...Ch. 1 - Prob. 71EQCh. 1 - Prob. 72EQCh. 1 - Prob. 73EQCh. 1 - Prob. 74EQCh. 1 - Prob. 75EQCh. 1 - Heterolytic cleavage of the CO bond to yield a...Ch. 1 - Prob. 77EQCh. 1 - Prob. 78EQCh. 1 - Prob. 79EQCh. 1 - Prob. 80EQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Select the correct lewis structure. [Ca-H] [H.][Ca:][H.] ○[H:]¯ [Ca]²+[H:]" [H-Ca-H] [H:][Ca]+arrow_forward___________ pairs: shared electrons in a Lewis structure; they can be represented by two dots or one line, NOT both!arrow_forwardQ5. Use your knowledge of formation of ionic compounds and the cross over method. Show how Ca3(PO4)2 and CaCO3 were formed. Load a picture of your working (2)arrow_forward
- Draw the Lewis-dot structure for C4 H10 below. Remember the Lewis-dot structure is the same asthe Expanded structure.arrow_forwardAn ionic bond forms when _________.a. atoms share electronsb. positively charged and negatively charged parts ofcovalent molecules attractc. ions with opposite electrical charges attractd. two atoms exchange protonsarrow_forwardPlease help with question 2 and 8. Thank you so much! 2. What is the molecular formula of your mononitrated reaction product? 8. Based on all of the data provided, what is the identity of the “G” group? Please NEATLY & CLEARLY draw its full Lewis structure in the space providedarrow_forward
- Draw Lewis Structures for the following: CH4 NH3 H20 SiF. NClsarrow_forwardР ed Lewis structure H-CC-H [H_H_O_H]* Η N_0_a: Is the proposed Lewis structure reasonable? Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:*arrow_forward。 A Lewis structure for the carbonate ion (CO¹³½³-) is shown, but incomplete. Complete the structure by adding in formal charges and non-bonding electrons. Then draw the other two major resonance structures that fit the octet rule. Add missing charges and non-bonding electrons. Select Draw Templates More Draw major resonance structure 2. Be sure to add charges and non-bonding electrons where appropriate. Select Draw Templates More / // /// C C || C Q 2Q Erase Draw major resonance structure 3. Be sure to add charges and non-bonding electrons where appropriate. Erase Select Draw Templates More / // /// C Erasearrow_forward
- ketch a Lewis structure for nitrite. One resonance form will be fine. The O-N-O bond angle in nitrite is close to ____________ ° and has a ____________ molecular geometry. Select one: 120, linear 109, bent 120,bent 120, trigonal pyramidal 180, lineararrow_forwardWrite answers to these using mental math (without a calculator). Based on the balanced equation: 4 Fe + 3 O2 → 2 Fe2O3 1. To form four units of Fe2O3 , a. How many atoms of Fe are used?____________ b. How many molecules of O2 are needed?____________arrow_forwardSC ! F1 Q @ 2 F2 W #3 Which structure has a formal charge of -1 on the carbon shown with a "C"? Note that the atom of interest in each case includes any hydrogens or lone pairs that are present. =C- (b) (c) (d) O a.. O b.. O c.. O d.. A Moving to another question will save this response. >> 20 F9 F10 F3 F4 E @ $ 4 R % F5 T MacBook Air F6 6 Y & 7 F7 U * 00 8 DII FB ( 9 ) 0 I P a F11arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY