
Concept explainers
(a)
Interpretation:
The indicated bond angle in the following compound should be predicted:
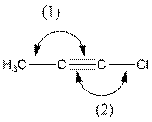
Concept Introduction:
The following table should be used while determining the bond around an atom.
| Number of groups | Number of atoms | Number of lone pairs | Shape | Bond angle |
| 2 | 2 | 0 | Linear |
|
| 3 | 3 | 0 | Trigonal planar |
|
| 4 | 4 | 0 | Tetrahedral |
|
| 4 | 3 | 1 | Trigonal pyramidal |
|
| 4 | 2 | 2 | Bent |
|
Answer to Problem 27P
Angle (1) and angle (2) both measures
Explanation of Solution
The given structure is as follows:
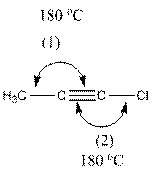
In the compound, triple bonded carbon is bonded to two other groups. If two groups surround an atom and no lone pair electron are present, the bond angle is
(b)
Interpretation:
The indicated bond angle in the following compound should be predicted:
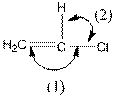
Concept Introduction:
The following table should be used while determining the bond angle around an atom.
| Number of groups | Number of atoms | Number of lone pairs | Shape | Bond angle |
| 2 | 2 | 0 | Linear |
|
| 3 | 3 | 0 | Trigonal planar |
|
| 4 | 4 | 0 | Tetrahedral |
|
| 4 | 3 | 1 | Trigonal pyramidal |
|
| 4 | 2 | 2 | Bent |
|
Answer to Problem 27P
Angle (1) and (2) both measures
Explanation of Solution
The compound is as follows:
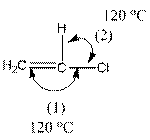
In the compound, three groups (hydrogen, carbon and chlorine) surround the double bonded carbon atom. And if three groups present surrounding an atom, the bond angle will be
(c)
Interpretation:
The indicated bond angle in the following compound should be predicted:
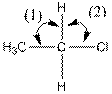
Concept Introduction:
The following table should be used while determining the bond angle around an atom.
| Number of groups | Number of atoms | Number of lone pairs | Shape | Bond angle |
| 2 | 2 | 0 | Linear |
|
| 3 | 3 | 0 | Trigonal planar |
|
| 4 | 4 | 0 | Tetrahedral |
|
| 4 | 3 | 1 | Trigonal pyramidal |
|
| 4 | 2 | 2 | Bent |
|
Answer to Problem 27P
Angle (1) and angle (2) both measures
Explanation of Solution
Given compound is as follows:
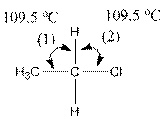
The carbon atom in the compound is surrounded by four groups (two hydrogen, one carbon and one chlorine). So, the angle between the bonds is
Want to see more full solutions like this?
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Which one of the following covalent compound structures is characterized by the SMALLEST bond angles between pendant hydrogen atoms? H₂O dihydrogen monoxide H-O: H H₂O CH4 NH3 CH₂ carbon tetrahydride H H-C-H H NH₂ nitrogen trihydride H-N-H -I Harrow_forward2. Identify the electron and molecular geometry of each indicated atoms in the compound below. HOarrow_forwardDraw correct line-bond structures for the following moleculesarrow_forward
- Give the line structure for each compound.arrow_forwardWrite the common (not systematic) name of each organic molecule. structure CH3 CH₂ CH3 CH3 CH-N-CH-CH3 CH3 CH₂ CH3 CH3 CH₂ CH₂-N-CH₂ - CH3 CH3 CH3—CH—NH—CH3 name 0 0 0arrow_forwardDraw VSEPR sketches for each compound below. Then, indicate whether the each of the following compounds is polar or nonpolar by drawing molecular dipole moments when appropriate. CHCl3 CH2Cl2 CCl4 H2O CH3CH2CH2CH2CH3arrow_forward
- Choose the two molecules that are constitutional isomers. m Compound 1 Compound 2 Compound 3 Compound 4 A) Compounds 1 and 2 B) Compounds 1 and 3 C) Compounds 2 and 3 D) Compounds 3 and 4 E) Compounds 2 and 4arrow_forwardWrite the common (not systematic) name of each organic molecule. structure CH₂ CH3 CH3-CH₂-CH₂-N-CH₂-CH₂-CH3 -— CH3 CH3 -CH₂CH₂-N-CH₂-CH₂-CH3 CH2−N−CH2−CH2–CH3 CH3—CH,—NH–CH3 name 0 0 0arrow_forwardThere are at least three different molecules with the formula C3H8O. Draw a Lewis structure for each possible constitutional isomer. Be careful not to duplicate any structures.arrow_forward
- Using VSEPR, predict the bond angles about the carbon and nitrogen atoms in each pair of contributing structure. In what way do these bond angles change from one contributing structure to the otherarrow_forward6. Describe concisely a chemical test to distinguish between the following pairs of compounds.arrow_forwardProvide the formula for each compound.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
