
Answer the following questions about levonorgestrel (trade name Plan B). Levonorgestrel interferes with ovulation, the release of an egg from an ovary, so it prevents pregnancy if taken within a few days of unprotected sex.
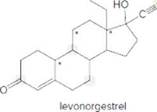
- Identify the
functional groups. - How many C’s does levonorgestrel contain?
- How many H’s are present at each C labeled with an asterisk (*)?
- Give the shape around each atom labeled in gray.
- Label all polar bonds.
- Is levonorgestrel soluble in an organic solvent?
- Is levonorgestrel soluble in water?
(a)
Interpretation:
The functional group in levonorgestral should be identified.
Concept Introduction:
Functional groups are the atoms or group of atoms that gives chemical properties to an organic compound and also chemical reactivity centre. Examples of functional groups are aldehyde, ketone, alcohol, alkene, alkyne etc.
Answer to Problem 84P
The functional groups present in the compound are alcohol, ketone, alkene and alkyne.
Explanation of Solution
Functional groups are the atoms or group of atoms that gives chemical properties to an organic compound and also chemical reactivity centre. Structures of different functional groups are as follows:
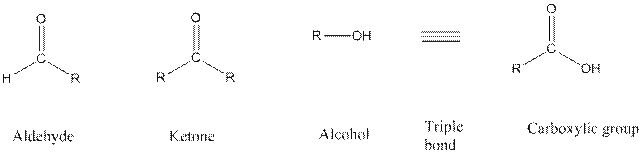
In the above functional group R represents an alkyl group.
In functional group ketone carbonyl carbon is bonded to two alkyl groups, in aldehyde carbonyl carbon is attached to one alkyl group and one hydrogen atom, in alcohol functional group an −OH is bonded to an alkyl group, in alkyne there is a triple bond between two carbon atoms and in alkene there is a double bond between two carbon atoms.
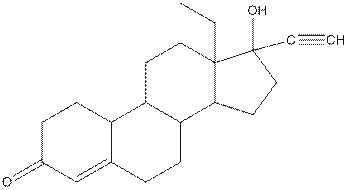
Levonorgestral structure is as follows:
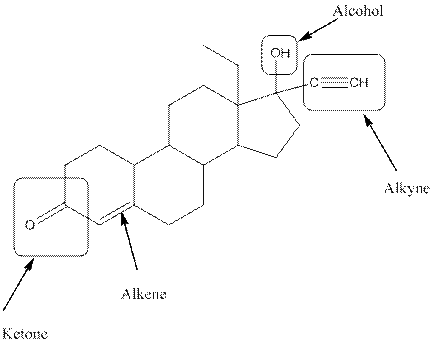
Observe the structure of Levonorgestral. As indicated in the above figure, four types of functional groups present that is, alkene, alkyne, alcohol and ketone. The functional groups present in the compound are alcohol, ketone, alkene and alkyne.
(b)
Interpretation:
The number of carbon atoms in the structure of levonorgestral should be determined.
Concept Introduction:
In skeletal structure the terminals represent methyl
Answer to Problem 84P
Twenty one carbon atoms present in the compound.
Explanation of Solution
The structure of Levonorgestral is as follows:
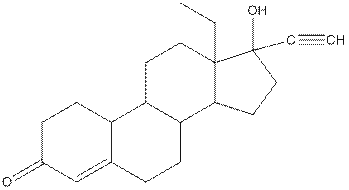
In skeletal structure the terminals represent methyl
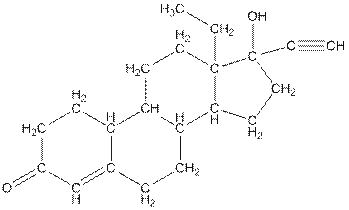
Hence, twenty one carbon atoms present in the compound.
(c)
Interpretation:
The number of hydrogen atoms present in the * carbon atoms in levonorgestral should be determined.
Concept Introduction:
In skeletal structure the terminals represent methyl
Answer to Problem 84P
Number of hydrogen atoms in * carbon atoms are three.
Explanation of Solution
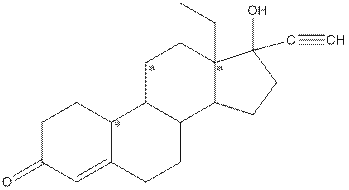
Complete structure of levonorgestral is as follows:
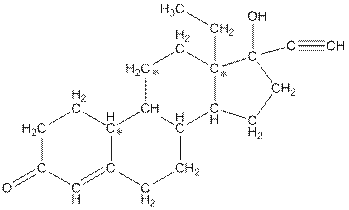
From the complete structure of Levonorgestral, it is clear that number of hydrogen atoms in * carbon atoms are three.
(d)
Interpretation:
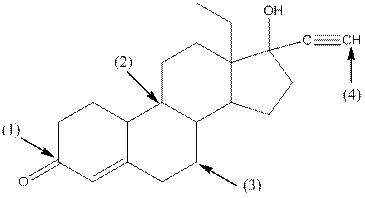
The shape around each indicated carbon atom in levonorgestral should be determined.
Concept Introduction:
The following table should be used while determining the shape around an atom.
| Number of groups | Number of atoms | Number of lone pairs | Shape | Bond angle |
| 2 | 2 | 0 | Linear | |
| 3 | 3 | 0 | Trigonal planar | |
| 4 | 4 | 0 | Tetrahedral | |
| 4 | 3 | 1 | Trigonal pyramidal | |
| 4 | 2 | 2 | Bent |
Answer to Problem 84P
Shape around carbon 1 is trigonal planar, shape around carbon 2 is tetrahedral, shape around carbon 3 is tetrahedral and shape around carbon 4 is linear.
Explanation of Solution
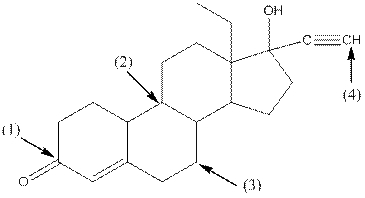
Given compound is as follows:
Complete structure of the compound is as follows:
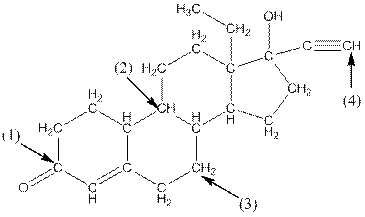
- Around C1, three atoms (two carbon and one oxygen) present. So, shape around carbon 1 is trigonal planar.
- Around C2, four atoms (three carbon and one hydrogen) present. So, shape around carbon 2 is tetrahedral.
- Around C3, four atoms (two carbon and two hydrogen) present. So, shape around carbon 3 is tetrahedral.
- Around C4, two atoms ( one carbon and one hydrogen) present. So, shape around carbon 4 is linear.
(e)
Interpretation:
All the polar bonds in levonorgestral should be labeled.
Concept Introduction:
The unequal sharing of valence electrons in a bond is called polar bond. Polar bond result when the bond formed between two atoms in which one atom is more electronegative than the other one. One example of polar bond is
Structure of HCl is as follows:

In
Answer to Problem 84P
The structure of levonorgestral with labeled all polar bonds is as follows:
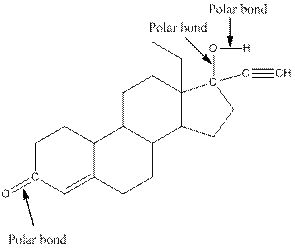
Explanation of Solution
In organic compound, most of the polar bonds formed between carbon and heteroatoms like oxygen, nitrogen, sulphur etc.
In the compound, three polar bonds present. One polar bond is between carbon and oxygen where oxygen is more electronegative than carbon. Second polar bond is between carbon and oxygen where oxygen is more electronegative than carbon and third polar bond is between oxygen and hydrogen where oxygen is more electronegative than hydrogen.
The structure of levonorgestral with labeled all polar bonds is as follows:
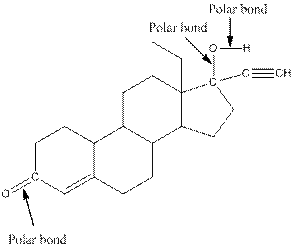
(f)
Interpretation:
Whether levonorgestral is soluble in organic solvent or not should be determined.
Concept Introduction:
A compound is soluble in organic solvent if the compound contains too many hydrocarbon bonds that is if it is non-polar.
Answer to Problem 84P
The compound levonorgestral will soluble in organic solvent.
Explanation of Solution
The complete structure of levonorgestral is as follows:
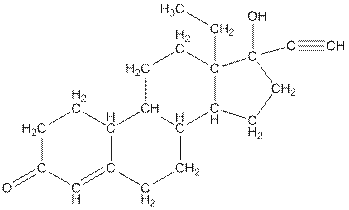
The compound levonorgestral has too many carbon hydrogen bonds. So, the compound will soluble in organic solvent.
(g)
Interpretation:
Whether levonorgestral is soluble in water or not should be determined.
Concept Introduction:
Water is a polar solvent. To dissolve in water the compound must be polar. Polar compound is that compound in which polar bonds are present. The unequal sharing of valence electrons in a bond is called polar bond. Polar bond result when the bond formed between two atoms in which one atom is more electronegative than the other one. One example of polar bond is
Structure of HCl is as follows:

In
Answer to Problem 84P
The compound levonorgestral is water soluble.
Explanation of Solution
Structure of levonorgestral is as follows:
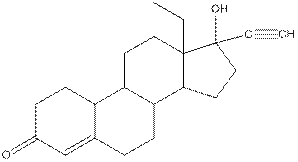
In the compound levonorgestral, two oxygen heteroatoms present. These heteroatoms can form hydrogen bonds with water molecule and hence, the compound is soluble in water.
Want to see more full solutions like this?
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- X. Circle and name each functional group in the following structure. H -OH cortisone acetate (active ingredient in steriod skin cream)arrow_forwardThe fact that sweet-tasting carbohydrates like table sugar are also high in calories has prompted the development of sweet, low-calorie alternatives, (a) Identify the functional groups in aspartame, the artificial sweetener in Equal, (b) Label all of the sites that can hydrogen bond to the oxygen atom of water, (c) Label all of the sites that can hydrogen bond with a hydrogen atom of water.arrow_forwardConsider the following two molecules and answer the following questions. Molecule A: НО C. CH-CH3 CH3 Molecule B: H;C-CH,- -C-0-CH3 a) What are all of the IMFS present in molecule A? b) What are all of the IMFS present in molecule B? c) Which would have the higher boiling point, molecule A or molecule B? d) What is the relationship between molecules A & B: structural isomers, geometric isomers, identical, or not related?arrow_forward
- Identify the functional group that appears in the following molecule that is known as spermine. Spermine is involved in cellular metabolism found in all eukaryotic cells. Crystals of spermine phosphate were first described in 1678, in human semen, by Anton van Leeuwenhoek. H,N. NH, Nitrile Ketone O Amine O Aldehyde O Amidearrow_forwardCH3CH2OH CH3SH CH3CH2CH3 Which compound can form hydrogen bonds? Which compound has the highest boiling point? Which compound has the lowest boiling point?arrow_forwardThe skeletal formula represents what type of alcohol? primary secondary tertiary quaternary none QUESTION 2 Thiols are strong- smelling compounds responsible for ________. skunky or bad smelling odors flowery odors sharp odors salty odors fruity odors QUESTION 3 The skeletal formula represents what type of alcohol? primary secondary tertiary quaternary none QUESTION 4 A phenol has an - OH group bonded to a(n) ________. tetrasubstituted carbon disubstituted carbon singly substituted or unsubstituted carbon trisubstituted carbon carbon. in a benzene ring QUESTION 5 The common name of CH 3— CH 2— O— CH 2— CH 3 is ________. 2-etherbutane diethyl ether dimethyl ether dibutyl ether butyl ether QUESTION 6 In the IUPAC naming system, a ketone is named by replacing the - e in the corresponding alkane name with ________. al ol one ene yne QUESTION 7 When a primary alcohol is strongly oxidized, the product is ________. an aldehyde an alkane another alcohol a carboxylic acid a…arrow_forward
 Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





