
Concept explainers
Show how each of the following compounds can be synthesized from cyclopentanol and any necessary organic or inorganic reagents. In many cases the desired compound can be made from one prepared in an earlier part of the problem.
trans-
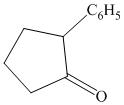
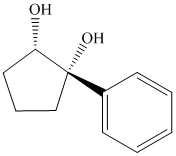
Interpretation:
Each of the given product is to be synthesized from cyclopentanol and necessary organic or inorganic reagents.
Concept introduction:
Alcohols can be prepared from a variety of reagents.
Reaction of Grignard reagents with carbonyl compounds produces the corresponding alcohols.
Grignard reagents also react with oxiranes to produce alcohols.
The allylic and benzylic carbon atoms are selectively brominated using NBS reagent.
Alcohols undergo dehydration in acidic medium, producing alkenes. These alkenes can be converted to diols using osmium tetra oxide.
Answer to Problem 23P
Solution:
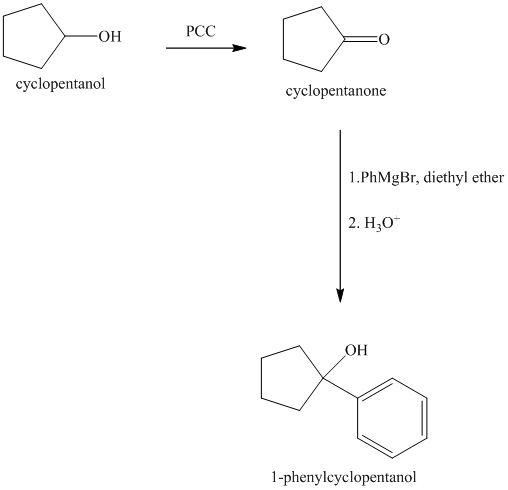
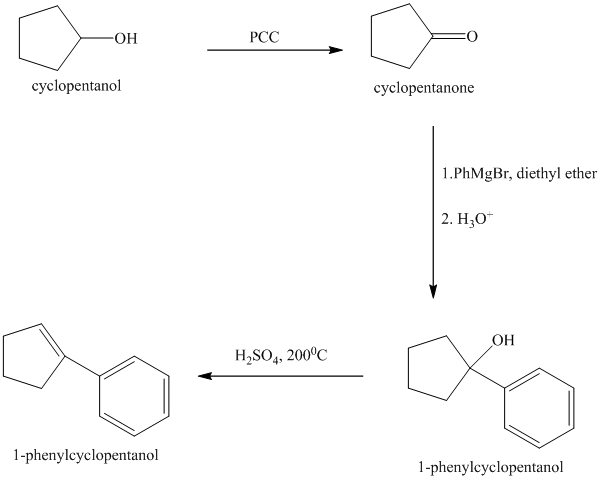
Explanation of Solution
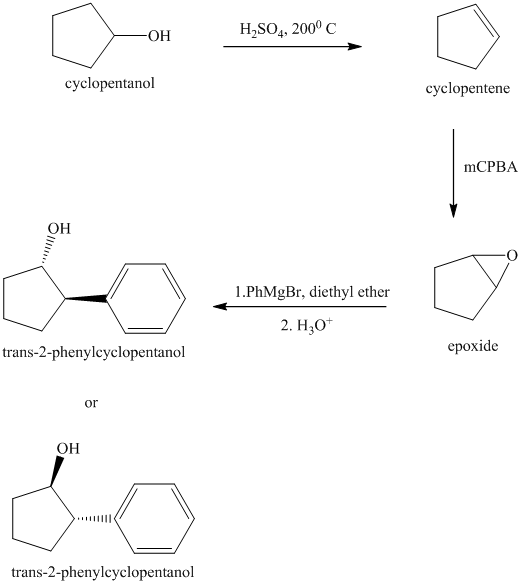
Synthesis of
The structure for cyclopentanol and
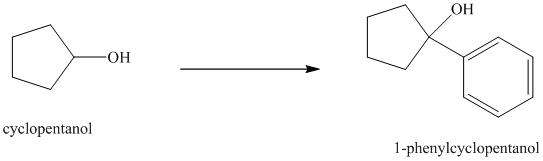
In

Synthesis of
The structure for cyclopentanol and
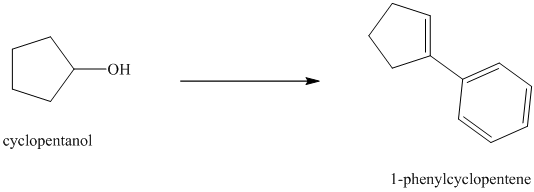
In
The sequence of reactions starting from cyclopentanol to yield the final given product

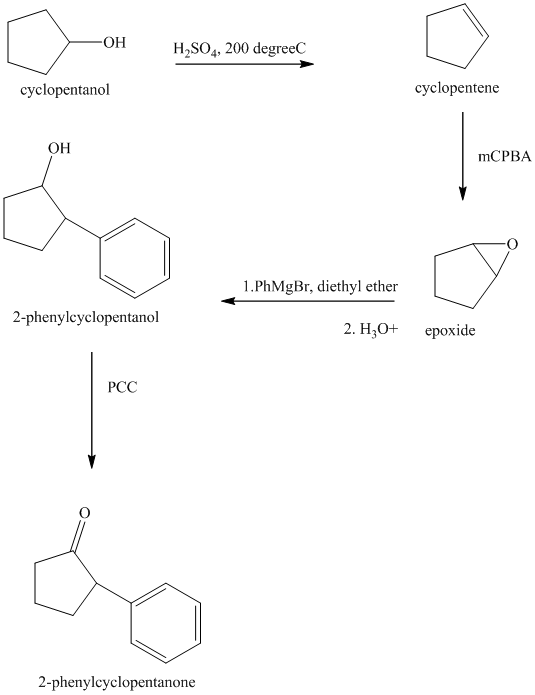
Synthesis of
The structure for cyclopentanol and
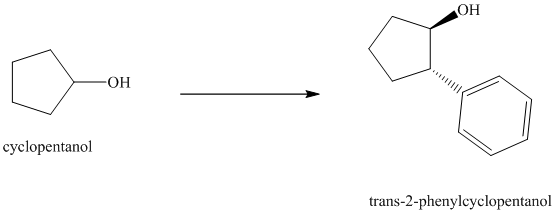
In
The sequence of reactions starting from cyclopentanol to yield the final given product

Synthesis of
The structure for cyclopentanol and
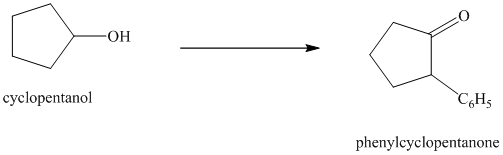
In
The sequence of reactions starting from cyclopentanol to yield the final given product
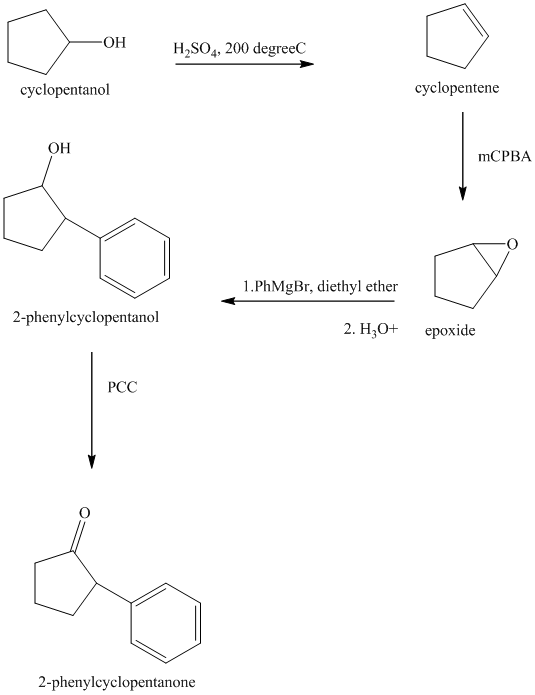
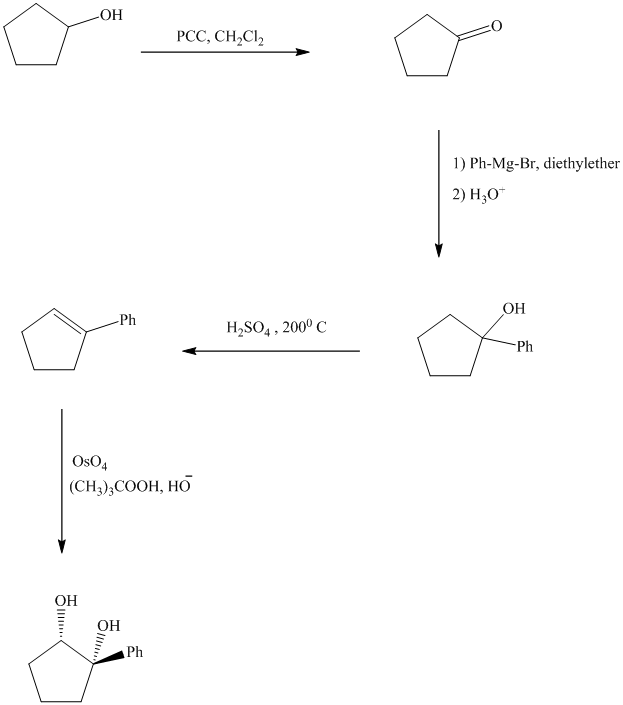
Synthesis of the given diol from cyclopentanol.
The structure for the given diol is as follows:
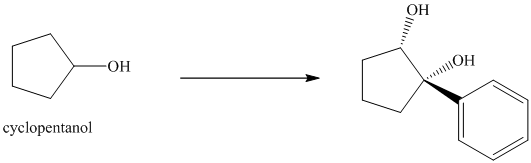
In the given diol, one phenyl ring and one hydroxyl group are attached to the same carbon of cyclopentane ring. The other hydroxyl group is attached to C2 position of cyclopentane ring.
Oxidation of the cyclopentanol will produce cyclopentanone. Reaction of this cyclopentanone with phenyl magnesium bromide will form a tertiary alcohol. Acid catalyzed dehydration of this tertiary alcohol will produce
The sequence of reactions starting from cyclopentanol to yield the final given product

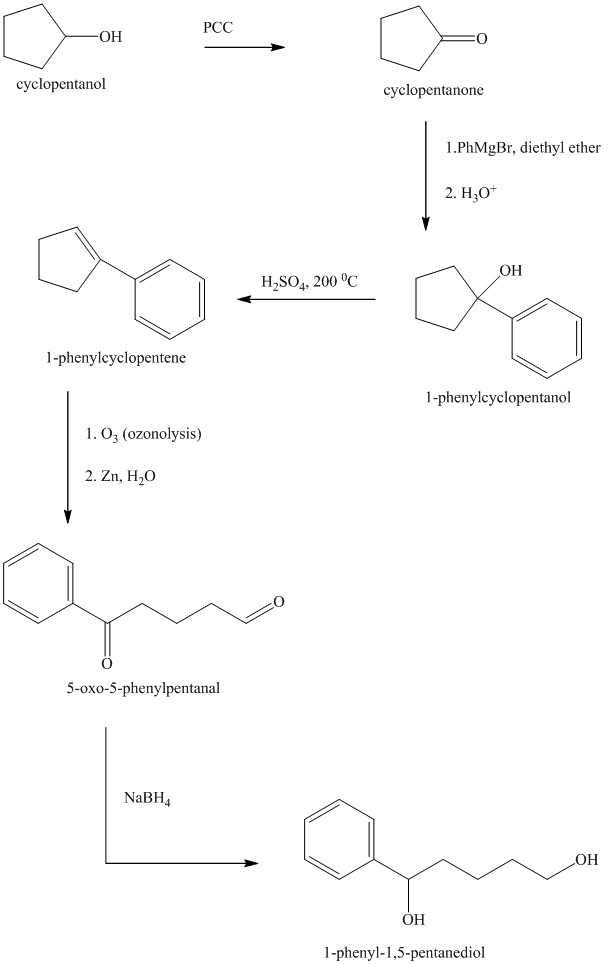
Synthesis of
The structure for cyclopentanol and
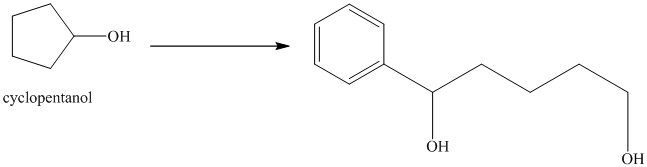
Cyclopentanol, when undergoes oxidation in presence of a mild oxidizing agent such as pyridinium chlorochromate in dichlormethane, the hydroxyl group turns to a carbonyl group and forms cyclopentanone. Cyclopentanone, when treated with Grignard reagent (PhMgBr) in the presence of diethyl ether with acidic workup, forms
The sequence of reactions is shown below.

Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry - Standalone book
- Using your reaction roadmaps as a guide, show how to convert propane into propyl propanoate. You must use propane as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forwardThe following molecule belongs to a class of compounds called enediols; each carbon of the double bond carries an OH group. Draw structural formulas for the -hydroxyketone and the -hydroxyaldehyde with which this enediol is in equilibrium.arrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-bromopropane and carbon dioxide into 4-propyl-4-heptanol. You must use 1-bromopropane and carbon dioxide as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forward
- Choose the best reagents to complete the following reactions.arrow_forwardAcetals are formed from the reaction of two alcohols with a carbonyl under acidic conditions. Acetal formation is faster with 1,2-ethanediol than with two methanol molecules. Choose the factor that explains the difference in reaction rates. A) The reaction with 1,2-ethanediol has a lower AH (enthalpy) of reaction. B) The reaction with 1,2-ethanediol has a higher AH (enthalpy) of reaction. C) The reaction with 1,2-ethanediol has a more favorable entropy of reaction.arrow_forwardIdentify the best reagents to complete the following reaction. HO, CIarrow_forward
- 1% 83- 80- 70- 60- 50- 40- 30 20- 10- -0- 4000 3622.47cm-1 3547.27cm-1 3430.08cm-1 3064.48cm-1 3500 3033.50cm-1 2999.56cm-1 2952.56cm-1 3000 2844.00cm-1 2500 1725.00cm-1 cm-1 2000 | 1601.55cm-1 1435.67cm-1 1500 966.45cm-1 1027.19cm-1 1071.40cm-1 1111.83cm-1 1000 500arrow_forwardIdentify the best reagents to complete the following reaction. Options are included.arrow_forwardChoose the CORRECT name for this compound. OCH3 O 2-Methoxy-5-phenyl-3-pentanone O 3-Methoxy-1-phenyl-2-butanone 4-Methoxy-1-phenyl-3-pentanone 2-Methoxy-4-phenyl-3-butanonearrow_forward
- Which of the following compounds will NOT undergo nucleophilic substitution reaction? isopropanol Benzoyl chloride propylene oxide butanonearrow_forwardFor each of the transformations given below, draw the structures of the named starting material, products and possible intermediates. State the necessary reagents and conditions in the reaction scheme. More than one step may be required. cyclohexylmethanol ethenylbenzene pent-1-ene 2-methylpentan-3-ol cyclohexanecarbaldehyde acetophenone 2-methylpenta-2-amine propanoic acid + propanone a) b)arrow_forwardChoose the correct product for the following reaction 4-methylcyclohexanone + Br2/H3O+ --> product? A 2,6-dibromo-4-methylcyclohexanol B 1-bromo-4-methylcyclohexanol C 2,6-dibromo-4-methylcyclohexanone D 2-bromo-4-methylcyclohexanol E 2-bromo-4-methylcyclohexanonearrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

