
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.SE, Problem 36MP
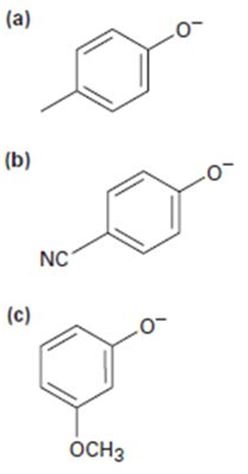
Phenols generally have lower pKa’s than aliphatic alcohols because of resonance stabilization with the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write down the common (not IUPAC) names of the organic molecules that would be released if this molecule were hydrolyzed:
CH2−O−C—(CH2);–CH=CH–CH2–CH=CH—(CH2)4—CH3
CH-O-C-(CH2)14-CH3
O
11
CH2−O−C— (CH2)14 — CH3
Separate each name with a comma. You will find useful information in the ALEKS Data resource.
1
010
Continue
O
a
X
000
Y
F8
F9
Submi
Indicate the carbanions that can be generated and which is
the most stable when the compound is treated with a base:
O
||
O
||
H3C-C-CH-CH₂-C-CH3
|
CO OEt
ethyl 2-acetyl-4-oxopentanoate
OChem help with IUPAC names involving Ph and Bn
The phenyl group (Ph-R, C6H5-R) can be formed by removing a hydrogen from benzene and attaching a substituent to where the hydrogen was removed.
The benzyl group (abbv. Bn), similar to the phenyl group, is formed by manipulating the benzene ring. In the case of the benzyl group, it is formed by taking the phenyl group and adding a CH2 group to where the hydrogen was removed. Its molecular fragment can be written as C6H5CH2-R, PhCH2-R, or Bn-R.
Please provide the IUPAC name for the following:
(Ph)2CHC(CH3)2CC(CH2)3CH(Bn)CHO
Chapter 17 Solutions
Organic Chemistry
Ch. 17.1 - Give IUPAC names for the following compounds:Ch. 17.1 - Prob. 2PCh. 17.2 - The following data for isomeric four-carbon...Ch. 17.2 - Rank the following substances in order of...Ch. 17.2 - Prob. 5PCh. 17.3 - Prob. 6PCh. 17.4 - What reagent would you use to accomplish each of...Ch. 17.4 - Prob. 8PCh. 17.5 - Prob. 9PCh. 17.5 - Prob. 10P
Ch. 17.5 - Use the reaction of a Grignard reagent with a...Ch. 17.6 - How would you carry out the following...Ch. 17.6 - What products(s) would you expect from dehydration...Ch. 17.7 - What alcohols would give the following products on...Ch. 17.7 - What products would you expect from oxidation of...Ch. 17.8 - TMS ethers can be removed by treatment with...Ch. 17.9 - Show the mechanism for the reaction of...Ch. 17.11 - Prob. 18PCh. 17.11 - When the 1HNMR spectrum of an alcohol is run in...Ch. 17.SE - Give IUPAC names for the following compounds:Ch. 17.SE - Draw the structure of the carbonyl compound(s)...Ch. 17.SE - Prob. 22VCCh. 17.SE - Prob. 23VCCh. 17.SE - Name and assign R or S stereochemistry to the...Ch. 17.SE - Evidence for the intermediate carbocations in the...Ch. 17.SE - Acid-catalyzed dehydration of 2,...Ch. 17.SE - Prob. 27MPCh. 17.SE - Treatment of the following epoxide with aqueous...Ch. 17.SE - Prob. 29MPCh. 17.SE - Prob. 30MPCh. 17.SE - Identify the type of substitution mechanism (SN1,...Ch. 17.SE - The conversion of 3 alcohols into alkenes under...Ch. 17.SE - Prob. 33MPCh. 17.SE - The trimethylsilyl (TMS) protecting group is one...Ch. 17.SE - When the alcohol below is treated with POCI3 and...Ch. 17.SE - Phenols generally have lower pKa’s than...Ch. 17.SE - Give IUPAC names for the following compounds:Ch. 17.SE - Draw and name the eight isomeric alcohols with...Ch. 17.SE - Prob. 39APCh. 17.SE - Named bombykol, the sex pheromone secreted by the...Ch. 17.SE - Carvacrol is a naturally occurring substance...Ch. 17.SE - What Grignard reagent and what carbonyl compound...Ch. 17.SE - What carbonyl compounds would you reduce to...Ch. 17.SE - What carbonyl compounds might you start with to...Ch. 17.SE - Prob. 45APCh. 17.SE - What products would you obtain from reaction of...Ch. 17.SE - Prob. 47APCh. 17.SE - How would you prepare the following compounds from...Ch. 17.SE - Prob. 49APCh. 17.SE - What products would you expect to obtain from...Ch. 17.SE - Prob. 51APCh. 17.SE - Propose structures for alcohols that have the...Ch. 17.SE - Propose a structure consistent with the following...Ch. 17.SE - The 1HNMR spectrum shown is that of...Ch. 17.SE - A compound of unknown structure gave the following...Ch. 17.SE - Propose a structure for a compound C15H24O that...Ch. 17.SE - Prob. 57APCh. 17.SE - Prob. 58APCh. 17.SE - Rank the following substituted phenols in order of...Ch. 17.SE - Benzvl chloride can be converted into benzaldehvde...Ch. 17.SE - Prob. 61APCh. 17.SE - Prob. 62APCh. 17.SE - Prob. 63APCh. 17.SE - Prob. 64APCh. 17.SE - Prob. 65APCh. 17.SE - Prob. 66APCh. 17.SE - Dehydration of trans-2-methylcyclopentanol with...Ch. 17.SE - 2, 3-Dimethyl-2, 3-butanediol has the common name...Ch. 17.SE - As a rule, axial alcohols oxidize somewhat faster...Ch. 17.SE - Prob. 70APCh. 17.SE - A problem often encountered in the oxidation of...Ch. 17.SE - Identify the reagents a-f in the Following scheme:Ch. 17.SE - Prob. 73APCh. 17.SE - Prob. 74APCh. 17.SE - Compound A, C8H10O, has the IR and 1H NMR spectra...Ch. 17.SE - Prob. 76APCh. 17.SE - Prob. 77AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 16-19 2-Me thy 1 propane (bp -12°C), 2-propanol (bp 82°C), and 2-propanamine (bp 32°C) all have approximately the same molecular weight, yet their boiling points are quite different. Explain the reason for these differences.arrow_forwardAn important step in one synthesis of carboxylic acids is the deprotonation of diethyl malonate and its alkyl-substituted derivative: Base CH;CH2O OCH,CH3 CH;CH,0 OCH2CH3 H2 Diethyl malonate Base CH;CH,0 °C `OCH,CH3 CH;CH,O OCH,CH3 R Alkyl substituted diethyl malonate NaOH can deprotonate diethyl malonate effectively, but NaOC(CH3)3 is typically used to deprotonate the alkyl-substituted derivative. Explain why.arrow_forwardPick the best IUPAC name for the following molecule: H3C-O CH3 CH3 H3C- O 2,4,5-trimethylheptanal O5-methoxy-2,4-dimethylheptanoic Acid O 5-methoxy-2,4-dimethylheptanal 5-methoxy-2,4-dimethylheptan-1-olarrow_forward
- Indicate the products A and B that are obtained in the following reactions: OH CH3 + HOCH2-CH₂OH + TSOH → A 1° NaH/THF A + →> B 2º C6H5-CH2Cl Briefly comment on each reaction.arrow_forwardDraw the skeletal structure of the ester formed when carboxylic acid CH₂CH(CH₂)CH₂CH₂CO₂H is treated with ethanol (CH₂CH₂OH) in the presence of H₂SO4. Click and drag to start drawing a structure. C™ X ں: G tu 2arrow_forwardWhat is the correct assignment of the names of the following ketones? H;C CH3 CH3 1 2 O 1 = acetone; 2 = phenol; 3 = benzaldehyde 1 = acetone; 2 = acetophenone; 3 = benzophenone %3D %3D O 1 = formaldehyde; 2 = benzaldehyde; 3 = acetophenone %3D O 1 = acetaldehyde; 2 = acetophenone; 3 = benzaldehyde %3D %3Darrow_forward
- You may want to reference (Pages 408 - 414) Section 12.3 while completing this problem. The compound frambinone has the taste of raspberries and has been used in weight loss. Its structure is HO |||-CH₂-CH, -C-CH3 ▾ Part A Identify the functional groups in frambinone. Check all that apply. ketone aromatic aldehyde ester ether phenol Submit Request Answerarrow_forwardFill in thhe blanks with the appropriate chemical compound for the following reactions: 1-methylcyclohexanol + phosphoric acid catalyst + heat --> + 2-methylcyclohexanol + phosphoric acid catalyst + heat --> 3-methylcyclohexane 3-methylcyclohexene methylenecyclohexane| 2-methylcyclohexene 1-methylcyclohexene 1-methylcyclohexane Next pag ge MacBook Airarrow_forwardHydration of aldehydes and ketones can be catalyzed by acid or base. Bases catalyze hydration by: protonating the carbonyl oxygen making the carbonyl group more electrophilic employing hydroxide ion, which is a better nucleophile than water making the carbonyl group less electrophilic shifting the equilibrium position of the reaction to favor productsarrow_forward
- Of the following substances. What are the products of the reaction between 2-ethyl-3-methyl butanoic acid and secbutyl alcohol? CH3 CH2-CH3 CH3 || cs) CH-CH-CH-COO-CH-CH2-CH3 y H₂O CH3 CH3-CH₂ CH3 11 tn) CH,—CH–CH–COO–CH2-CH-CH3 y H2O CH3 -CH-CH₂-CI CH3 CH₂-CH3 11 qy) CH-CH-CH-COO-CH-CH₂-CH3 y H₂ CH3 CH3-CH₂ CH3 11 da) CH–CH–CH–COO–CH2CH-CH3 y Hàarrow_forwardHow would you convert benzoic acid (C6H5CO2H) to each compound?arrow_forwardCH,CH,CH,-OH Draw the structure of the product expected when each of the following alcohols is reacted with a sulfuric acid catalyst au the temperature indicated. H,SO, CH,-CH-CH, 180°C a. OH H,SO, CH, — CH—СH,—ОН 180°C b. CH; H,SO, CH-CH-OH 140°C с. CH, CH,-CH-CH,-CH, H,SO, d. 140°C d. OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY